Disulfiram-d20
Disulfiram-d20
CAS: 1216403-88-1
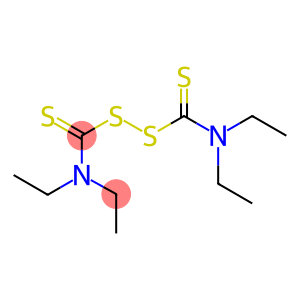
Molecular Formula: C10H20N2S4
Disulfiram-d20 - Names and Identifiers
| Name | Disulfiram-d20 |
| Synonyms | Disulfiram-d20 (Tetraethyl-d20)thioperoxydicarbonic DiaMide |
| CAS | 1216403-88-1 |
Disulfiram-d20 - Physico-chemical Properties
| Molecular Formula | C10H20N2S4 |
| Molar Mass | 296.52 |
| Melting Point | 51-58°C |
| Storage Condition | Refrigerator |
Disulfiram-d20 - Reference
| Reference Show more | 1. Feng Bing-yu, Zhu Yuan-gang, Wei Liang, etc. Inhibitory effect of artemisinin-β-Cyclodextrin Inclusion Complex combined with disulfiram on liver metastasis of colorectal cancer in vivo [J]. Specialty research, 2020, 042(001):6-10. |
Disulfiram-d20 - Introduction
Disulfiram-d20 is a heavy Proton substitute, its chemical formula is C10H20N2S2O-d20. It is a double proton substitute for aniline dithionyl compounds, similar to the common Disulfiram, but substituting two hydrogen atoms for heavy deuterium atoms.
The main use of Disulfiram-d20 is to study and analyze the metabolism and efficacy of the drug diulfiram, especially for pharmacokinetic studies through nuclear magnetic resonance technology (NMR). It can be used to determine the distribution, metabolism and excretion pathways of Disulfiram in the body, as well as to evaluate drug interactions with other substances.
The preparation method of Disulfiram-d20 is similar to that of Disulfiram, which is obtained by reacting aniline with dithionyl chloride and then metabolizing with heavy water (sodium deuterium oxide). This process ensures the production of Disulfiram-d20, I .e., a D2 substitute containing the heavy dehydrosulfates.
Regarding safety information, Disulfiram-d20 is a relatively safe compound when used correctly. However, it is still a toxic compound and requires safe laboratory practices. Appropriate personal protective measures such as gloves, lab clothes and goggles are required when handling and using Disulfiram-d20. In addition, should avoid inhalation of its dust or vapor, avoid skin contact or ingestion. In case of accidental exposure or inhalation, seek medical help immediately.
The main use of Disulfiram-d20 is to study and analyze the metabolism and efficacy of the drug diulfiram, especially for pharmacokinetic studies through nuclear magnetic resonance technology (NMR). It can be used to determine the distribution, metabolism and excretion pathways of Disulfiram in the body, as well as to evaluate drug interactions with other substances.
The preparation method of Disulfiram-d20 is similar to that of Disulfiram, which is obtained by reacting aniline with dithionyl chloride and then metabolizing with heavy water (sodium deuterium oxide). This process ensures the production of Disulfiram-d20, I .e., a D2 substitute containing the heavy dehydrosulfates.
Regarding safety information, Disulfiram-d20 is a relatively safe compound when used correctly. However, it is still a toxic compound and requires safe laboratory practices. Appropriate personal protective measures such as gloves, lab clothes and goggles are required when handling and using Disulfiram-d20. In addition, should avoid inhalation of its dust or vapor, avoid skin contact or ingestion. In case of accidental exposure or inhalation, seek medical help immediately.
Last Update:2024-04-09 20:52:54
Supplier List
Product Name: Disulfiram-d20 Request for quotation
CAS: 1216403-88-1
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 1216403-88-1
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Product Name: Disulfiram-d20 Visit Supplier Webpage Request for quotation
CAS: 1216403-88-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 1216403-88-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: Disulfiram-d20 Request for quotation
CAS: 1216403-88-1
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 1216403-88-1
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Product Name: Disulfiram-d20 Visit Supplier Webpage Request for quotation
CAS: 1216403-88-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 1216403-88-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History

