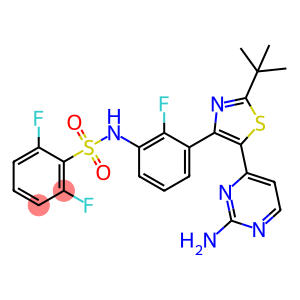
Dabrafenib
Dabrafenib
CAS: 1195765-45-7
Molecular Formula: C23H20F3N5O2S2
Dabrafenib - Names and Identifiers
Dabrafenib - Physico-chemical Properties
| Molecular Formula | C23H20F3N5O2S2 |
| Molar Mass | 519.56 |
| Density | 1.443 |
| Melting Point | 214-216oC |
| Boling Point | 653.7±65.0 °C(Predicted) |
| Solubility | Soluble in DMSO (up to 30 mg/ml with warming), or in Ethanol (up to 1 mg/ml with warming). |
| Appearance | White solid. |
| Color | Off-white |
| pKa | 6.62±0.10(Predicted) |
| Storage Condition | -20°C |
| Stability | Stable for 1 year from date of purchase as supplied. Solutions in DMSO or ethanol may be stored at -20°C for up to 3 months. |
| In vitro study | Dabrafenib is selective for Raf kinases and is 400-fold more active against B- Raf than 91% of the other kinases tested. Dabrafenib inhibits B- Raf V600E kinase, resulting in decreased ERK phosphorylation and inhibition of cell proliferation, cell arrest in G1 phase in cancer cells that specifically encode mutated B- Raf V600E. |
| In vivo study | Dabrafenib (oral) inhibits the growth of B- RafV600E mutated melanoma (A375P) and colon cancer (Colo 205) in immunocompromised mice. Dabrafenib (oral) also inhibits tumor growth. |
Dabrafenib - Risk and Safety
| HS Code | 29350090 |
Dabrafenib - Preparation solution concentration reference
| 1mg | 5mg | 10mg | |
|---|---|---|---|
| 1 mM | 1.925 ml | 9.624 ml | 19.247 ml |
| 5 mM | 0.385 ml | 1.925 ml | 3.849 ml |
| 10 mM | 0.192 ml | 0.962 ml | 1.925 ml |
| 5 mM | 0.038 ml | 0.192 ml | 0.385 ml |
Last Update:2024-01-02 23:10:35
Dabrafenib - Reference Information
| melanoma drug | Dabrafenib (Dabrafenib) is produced by GlaxoSmithKline (GSK) A drug developed for the treatment of metastatic melanoma is a BRAF inhibitor cancer drug, which is used in the form of mesylate. The former name is GSK2118436, and the trade name is Tafinlar. on 29-application for marketing. Tafinlar is approved for the treatment of unresectable melanomas (tumors that cannot be surgically removed) and metastatic melanomas (tumors that have spread to other organs in the body) carrying the BRAF V600E mutation, it cannot be used to treat wild-type BRAF melanoma. Mekinist is used to treat unresectable or metastatic melanoma with BRAF V600E or V600K mutations, and Mekinist should not be used to treat melanoma patients who have previously used BRAF inhibitors. The combination of these two drugs is considered to have a more effective and lasting therapeutic effect on melanoma, and the combination of the two drugs is also considered to be the main commercial opportunity for the two drugs. |
| synthesis method | The key step in the synthesis of Dabrafenib is the construction of 1, 3-thiazole ring, it is generally derived from direct cyclization of thioamides as 1,3-nucleophiles and α-carbonyl halides as 1,2-amphiphilic reagents. Sulfonyl chloride 1 and aniline 2 gave sulfonamide 3 under basic conditions. Methylpyrimidine 4 with non-nucleophilic strong base LiHMDS to remove the acidic proton on the methyl group after reaction with 3 to obtain 5, the latter with NBS alpha-bromination to obtain 1,2-amphiphilic reagent 6,6 is further reacted with 1,3-amphiphilic nucleophile 7 to obtain ring closure 8, and then reacted with ammonia water to obtain Dabrafenib. FIG. 1 shows the synthetic scheme of Dabrafenib. |
| biological activity | Dabrafenib (GSK2118436) is a mutant BRAFV600 specific inhibitor with an IC50 of 0.8 nM, the B- Raf(wt) and c-Raf effects were 4 and 6 times lower. Dabrafenib (GSK2118436, GSK2118436A) is a mutant BRAFV600 specific inhibitor with an IC50 of 0.7 nM in a cell-free assay, acting on B- Raf(wt). And c-Raf effect were 7 and 9 times lower, respectively. |
| Target | Value |
| B-Raf (V600E) (Cell-free assay) | 0.7 nM |
| B-Raf (Cell-free assay) | 5.2 nM |
| C-Raf (Cell-free assay) | 6.3 nM |
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: dabrafenib Visit Supplier Webpage Request for quotationCAS: 1195765-45-7
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: Dabrafenib Visit Supplier Webpage Request for quotationCAS: 1195765-45-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Dabrafenib Request for quotation
CAS: 1195765-45-7
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 1195765-45-7
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Spot supply
Product Name: Dabrafenib (GSK2118436) Visit Supplier Webpage Request for quotationCAS: 1195765-45-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: dabrafenib Visit Supplier Webpage Request for quotationCAS: 1195765-45-7
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: Dabrafenib Visit Supplier Webpage Request for quotationCAS: 1195765-45-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Dabrafenib Request for quotation
CAS: 1195765-45-7
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 1195765-45-7
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Spot supply
Product Name: Dabrafenib (GSK2118436) Visit Supplier Webpage Request for quotationCAS: 1195765-45-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



