DIALLYL TEREPHTHALATE
TEREPHTHALIC ACID DIALLYL ESTER
CAS: 1026-92-2
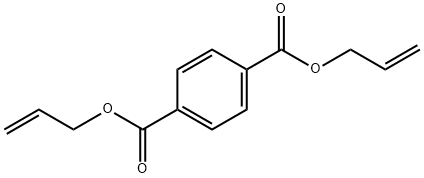
Molecular Formula: C14H14O4
DIALLYL TEREPHTHALATE - Names and Identifiers
| Name | TEREPHTHALIC ACID DIALLYL ESTER |
| Synonyms | DIALLYL TEREPHTHALATE Terephthalic acid diallyl p-Phthalic acid, diallyl ester TEREPHTHALIC ACID DIALLYL ESTER |
| CAS | 1026-92-2 |
| EINECS | 213-835-2 |
DIALLYL TEREPHTHALATE - Physico-chemical Properties
| Molecular Formula | C14H14O4 |
| Molar Mass | 246.26 |
| Density | 1,121 g/cm3 |
| Boling Point | 176-178°C 8mm |
| Flash Point | 171°C |
| Storage Condition | 2-8℃ |
| Refractive Index | 1.526 |
| MDL | MFCD00080462 |
DIALLYL TEREPHTHALATE - Risk and Safety
| UN IDs | 2810 |
DIALLYL TEREPHTHALATE - Preparation Method of diallyl terephthalate
Application (patent) number:
CN201810814882.3
application date:
2018.07.24
Public/Announcement Number:
CN108863778A
applicant (patent):
Shouguang Noreng Chemical Co., Ltd.
inventor:
Li Qingshan , Liu Xiaocheng , bright Wang
National and provincial code:
Shandong; 37
Abstract:
The present invention relates to a method for preparing diallyl terephthalate. Dimethyl terephthalate, allyl alcohol, calcium methoxide and/or calcium ethanol are added into a reactor, and stirring is started, temperature rise to 90~145 ℃, distillation column top temperature between 60~95 ℃, insulation reaction, sampling and testing, dimethyl terephthalate content is less than 0.5% stop heating; Cooling to below 100 ℃, add water, acid, continue to cool to 60 ° C., static stratification; Take the lower layer of water, layered, heated vacuum distillation to remove a small amount of water, to obtain diallyl terephthalate. The method greatly improves the efficiency of transesterification reaction, shortens the time of transesterification reaction, does not need decolorization treatment, the product has low color, improves the product quality, and the catalyst separation is simple, rapid and thorough, the cost of catalyst treatment is reduced; The overall process is more simplified, more suitable for advanced operation methods such as automated production, and the degree of automation of the overall process is improved.
stowed
sovereignty:
1. A preparation method of diallyl terephthalate, characterized in that the preparation method comprises the following steps:(1) Adding dimethyl terephthalate, allyl alcohol and a catalyst in a reactor, and stirring is turned on, and the temperature is raised to 90~145 °c for reaction, distillation column top temperature between 60~95 ℃, heat preservation reaction, sampling detection, stop heating when dimethyl terephthalate content is less than 0.5%;(2) cooling to below 100 ℃, add water, acid, the temperature was lowered to 60 ° C., and the mixture was allowed to stand and delaminated;(3) the lower layer was washed with water and delaminated, and a small amount of water was removed by heating and vacuum distillation to obtain diallyl terephthalate.
DIALLYL TEREPHTHALATE - Synthesis of diallyl terephthalate catalyzed by supported silicomolybdate
Author:
Fu Junke , fan Qiping , to Yulian , Chen Hong
Abstract:
diallyl terephthalate is an important monomer for the synthesis of optical materials, and the resin obtained by self-polymerization or copolymerization with other monomers is widely used in the manufacture of optical lenses, optical disks, optical fibers, optoelectronic devices, etc. There are few reports on the synthesis method of diallyl terephthalate. One of the published methods is to use benzene or toluene as a water-carrying agent and catalyze the reaction of terephthalic acid and propylene alcohol with concentrated sulfuric acid to prepare the target product, the second method is the reaction of sodium terephthalate with propenyl chloride in the presence of CuCl. In this paper, these methods are verified, and it is found that in the direct esterification method, because benzene or toluene is azeotropic with water and allyl alcohol, the proportion of allyl alcohol in the azeotrope is large, and the propylene alcohol is miscible with water, therefore, the loss of allyl alcohol in the process of carrying water is great. The main disadvantage of sodium salt synthesis is that the solubility of sodium terephthalate in water is very small, the reaction rate is very slow, and the hydrolysis rate of allyl chloride in the alkaline system is very fast, so it needs many times excessive amount of allyl chloride, resulting in waste of raw materials, moreover, the wastewater containing propylene alcohol has serious environmental pollution, so these two methods do not have the value of scale-up production. In this paper, under the catalysis of Heteropoly acid, diallyl terephthalate was prepared by transesterification of allyl alcohol with dimethyl terephthalate. The methanol was distilled off by fractional distillation, and the molar ratio of allyl alcohol to dimethyl terephthalate was 2.5:1. Compared with other types of catalysts, the catalytic activity of heteropolyacid is higher, and the polyaniline supported silicomolybdic acid catalyst needs only 1% of the raw material weight ratio, the reaction was quantitatively completed within 4HR, and the catalyst could be recycled. The method is simple and efficient, low pollution and low cost, is a green synthesis method to meet the needs of industrial production.
stowed
Key words:
load silicomolybdic acid synthesis diallyl terephthalate
meeting name:
The 7th National Symposium on research and progress of trace elements
meeting place:
Sanya, Hainan, China
DIALLYL TEREPHTHALATE - Study on processing properties and mechanical properties of poly (vinyl chloride)/diallyl phthalate polymer
from hownet
Author:
Chen Jihua , Wang Wei , skin red ,CHEN,Jihua,WANG,Wei,PI,Hong,State
Abstract:
The processability of poly (vinyl chloride)/diallyl phthalate (PVC/DAP) polymer was studied by torque rheometer. The effect of DAP content on the processability of PVC was discussed. The results showed that with the increase of DAP content, the plasticizing time of PVC/DAP decreased, the equilibrium torque and peak torque decreased, and the equilibrium temperature decreased. The tensile strength and elongation at break increased.Key words:
PVC diallyl phthalate processability mechanical properties
DOI:
CNKI:SUN:ZGSU.0.2017-03-006
cited:
year:
2017
DIALLYL TEREPHTHALATE - Synthesis of diallyl phthalate catalyzed by P-toluene sulfonic acid
from Palm Bridge research
Author:
Sun Yingjie , Chen Dijuan , Zhang Guangyou
Abstract:
diallyl phthalate was synthesized from phthalic anhydride and allyl alcohol with p-toluene sulfonic acid as catalyst. The molar ratio of phthalic anhydride to allyl alcohol is 1:4, the mass of p-toluenesulfonic acid is 13% of the mass of phthalic anhydride, and the yield of diallyl phthalate can reach 95.7% after refluxing for 16H.
Key words:
diallyl phthalate p-toluenesulfonic acid catalyst synthesis
year:
2009
Supplier List
CAS: 1026-92-2
Tel: +86 19849939632
Email: 2470479589@qq.com
Mobile: +86 19849939632
QQ: 2470479589
Wechat: +86 19849939632
CAS: 1026-92-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 1026-92-2
Tel: 15335994747
Email: 3521670276@qq.com
Mobile: 15335994747
QQ: 3521670276
Wechat: A2020071098
CAS: 1026-92-2
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 1026-92-2
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 1026-92-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 1026-92-2
Tel: +86 19849939632
Email: 2470479589@qq.com
Mobile: +86 19849939632
QQ: 2470479589
Wechat: +86 19849939632
CAS: 1026-92-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 1026-92-2
Tel: 15335994747
Email: 3521670276@qq.com
Mobile: 15335994747
QQ: 3521670276
Wechat: A2020071098
CAS: 1026-92-2
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 1026-92-2
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 1026-92-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025





