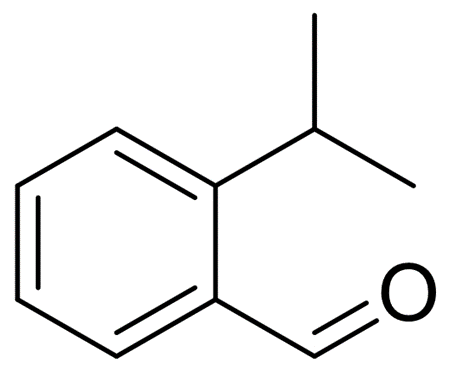
Benzaldehyde,2-(1-Methylethyl)-
Benzaldehyde, 2-(1-methylethyl)-
CAS: 6502-22-3
Molecular Formula: C10H12O
Benzaldehyde,2-(1-Methylethyl)- - Names and Identifiers
Benzaldehyde,2-(1-Methylethyl)- - Physico-chemical Properties
| Molecular Formula | C10H12O |
| Molar Mass | 148.2 |
| Density | 0.980 |
| Boling Point | 104°C/12.2mm |
| Flash Point | 91℃ |
| Storage Condition | under inert gas (nitrogen or Argon) at 2-8°C |
Benzaldehyde,2-(1-Methylethyl)- - Risk and Safety
| Hazard Symbols | Xi - Irritant |
| Risk Codes | R37/38 - Irritating to respiratory system and skin. R41 - Risk of serious damage to eyes R51 - Toxic to aquatic organisms |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S39 - Wear eye / face protection. |
Benzaldehyde,2-(1-Methylethyl)- - Introduction
Benzaldehyde, 2-(1-methyllethyl)-(Benzaldehyde, 2-(1-methyllethyl)-) is an organic compound with the chemical formula C10H12O.
Nature:
-Appearance: colorless liquid
-Smell: Bitter Almond Flavor (Almond Flavor)
-solubility: soluble in ethanol, ether, water, slightly soluble in sodium carbonate solution
Use:
-Spices: Benzaldehyde, 2-(1-methyllethyl)-can be used as a spice in food, beverage and perfume, giving the product an almond smell
-Raw material: It can be used as a starting material for the synthesis of other organic compounds, such as the synthesis of drugs and pesticides
Preparation Method:
Benzaldehyde, the preparation of 2-(1-methyllethyl)-is usually obtained by the hydrogenation reaction of almond acid (mandelonitrile). Specific steps are as follows:
1. the reaction of amygdylic acid with acetic anhydride to form mandelic acid acetate (mandelate acetate).
2. Hydrolysis of mandelic acid acetate to obtain mandelic acid.
3. almond acid generates Benzaldehyde, 2-(1-methyllethyl)-by heating decarboxylation reaction-.
Safety Information:
Benzaldehyde, 2-(1-methyllethyl)-is generally considered safe for humans under normal conditions of use. However, the following safety precautions should be noted:
-Avoid contact with skin, eyes and mucous membranes. In case of contact, rinse immediately with plenty of water.
-avoid breathing its vapor or moisture, ventilation measures can be taken.
-should be stored away from the fire and oxidant, stored in a cool, dry place.
Nature:
-Appearance: colorless liquid
-Smell: Bitter Almond Flavor (Almond Flavor)
-solubility: soluble in ethanol, ether, water, slightly soluble in sodium carbonate solution
Use:
-Spices: Benzaldehyde, 2-(1-methyllethyl)-can be used as a spice in food, beverage and perfume, giving the product an almond smell
-Raw material: It can be used as a starting material for the synthesis of other organic compounds, such as the synthesis of drugs and pesticides
Preparation Method:
Benzaldehyde, the preparation of 2-(1-methyllethyl)-is usually obtained by the hydrogenation reaction of almond acid (mandelonitrile). Specific steps are as follows:
1. the reaction of amygdylic acid with acetic anhydride to form mandelic acid acetate (mandelate acetate).
2. Hydrolysis of mandelic acid acetate to obtain mandelic acid.
3. almond acid generates Benzaldehyde, 2-(1-methyllethyl)-by heating decarboxylation reaction-.
Safety Information:
Benzaldehyde, 2-(1-methyllethyl)-is generally considered safe for humans under normal conditions of use. However, the following safety precautions should be noted:
-Avoid contact with skin, eyes and mucous membranes. In case of contact, rinse immediately with plenty of water.
-avoid breathing its vapor or moisture, ventilation measures can be taken.
-should be stored away from the fire and oxidant, stored in a cool, dry place.
Last Update:2024-04-09 20:45:29
Supplier List
Product Name: Benzaldehyde, 2-(1-methylethyl)- Request for quotation
CAS: 6502-22-3
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 6502-22-3
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Spot supply
Product Name: 2-Iso-Propylbenzaldehyde Visit Supplier Webpage Request for quotationCAS: 6502-22-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-ISO-PROPYLBENZALDEHYDE Request for quotation
CAS: 6502-22-3
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 6502-22-3
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Spot supply
Product Name: 2-Iso-Propylbenzaldehyde Visit Supplier Webpage Request for quotationCAS: 6502-22-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: Benzaldehyde, 2-(1-methylethyl)- Request for quotation
CAS: 6502-22-3
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 6502-22-3
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Spot supply
Product Name: 2-Iso-Propylbenzaldehyde Visit Supplier Webpage Request for quotationCAS: 6502-22-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-ISO-PROPYLBENZALDEHYDE Request for quotation
CAS: 6502-22-3
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 6502-22-3
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Spot supply
Product Name: 2-Iso-Propylbenzaldehyde Visit Supplier Webpage Request for quotationCAS: 6502-22-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



