BROMOSULFONPHTHALEIN
Sulfobromophthalein sodium
CAS: 123359-42-2
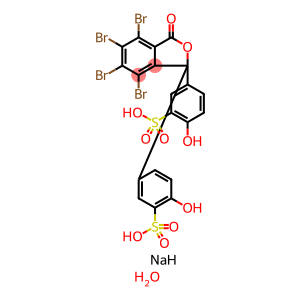
Molecular Formula: C20H13Br4NaO11S2
BROMOSULFONPHTHALEIN - Names and Identifiers
| Name | Sulfobromophthalein sodium |
| Synonyms | BROMO SULPHALEIN BROMOSULFOPHTHALEIN bromsulfalein sodium BROMOSULFONPHTHALEIN bromosulfalein sodium Sulfobromophthalein sodium BROMOSULPHALEIN DISODIUM SALT BROMOSULFOPHTHALEIN SODIUM SALT SULFORBROMOPHTHALEINSODIUMHYDRATE PHENOLTETRABROMOPHTHALEIN-3',3''-DISULFONIC ACID DISODIUM SALT 3,3'-(4,5,6,7-tetrabromo-3-oxo-1(3h)-isobenzofuranylidene)bis(6-hydroxy-benzenesulfonic acid) disodium salt |
| CAS | 123359-42-2 |
| EINECS | 626-376-1 |
| InChI | InChI=1/C20H10Br4O10S2.2Na.H2O/c21-15-13-14(16(22)18(24)17(15)23)20(34-19(13)27,7-1-3-9(25)11(5-7)35(28,29)30)8-2-4-10(26)12(6-8)36(31,32)33;;;/h1-6,25-26H,(H,28,29,30)(H,31,32,33);;;1H2/q;2*+1;/p-2 |
BROMOSULFONPHTHALEIN - Physico-chemical Properties
| Molecular Formula | C20H13Br4NaO11S2 |
| Molar Mass | 836.04 |
| Melting Point | >235°C (dec.) |
| Solubility | DMSO (Slightly), Methanol (Slightly), Water (Slightly) |
| Appearance | Crystalline Powder |
| Color | White |
| Storage Condition | room temp |
| Physical and Chemical Properties | WGK Germany:3 RTECS:SM8395000 |
BROMOSULFONPHTHALEIN - Risk and Safety
| Hazard Symbols | Xn - Harmful |
| Risk Codes | R42/43 - May cause sensitization by inhalation and skin contact. |
| Safety Description | S22 - Do not breathe dust. S36/37 - Wear suitable protective clothing and gloves. S45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) |
| WGK Germany | 3 |
| RTECS | SM8395000 |
| HS Code | 29329900 |
BROMOSULFONPHTHALEIN - Introduction
Sulfobromophthalein sodium (sodium sulfobromophthalein) is an organic compound, chemical formula is C20H10Br4Na2O5S, belongs to the phenol compound. It is a yellow crystalline powder, odorless, soluble in water. Sulfobromophthalein sodium is a biological dye that is often used as a tool to assess the metabolic function of bilirubin.
The main use of sodium bromide is to measure liver function, especially to detect the excretion of bilirubin. After oral administration of sodium sulfobromide, it is converted to a colorless metabolite in the liver and excreted into the bile. The ability of liver to metabolize bilirubin and the patency of the biliary tract can be assessed by measuring changes in the concentration of sodium sulfobromide in urine.
the preparation method of sodium sulfobromophthalein is generally to obtain sulfonic acid phenol through sulfonation reaction of phenol, then to obtain sulfonic acid bromophenol through bromination reaction, and finally to obtain sodium sulfobromophthalein through condensation reaction with catechol.
There are some safety information to note when using sodium sulfobromophthalein. It may cause irritation to the skin, eyes and respiratory system when touched, inhaled or ingested. Therefore, it is necessary to wear appropriate protective equipment to avoid direct contact when using sodium sulfobromophthalide. At the same time, keep it in a dry, cool, well-ventilated place, away from fire sources and other combustible substances. In the event of an accident, clean the affected area immediately and seek medical help. Finally, the use of sodium sulfobromide should follow the relevant national and regional regulations and safety procedures.
The main use of sodium bromide is to measure liver function, especially to detect the excretion of bilirubin. After oral administration of sodium sulfobromide, it is converted to a colorless metabolite in the liver and excreted into the bile. The ability of liver to metabolize bilirubin and the patency of the biliary tract can be assessed by measuring changes in the concentration of sodium sulfobromide in urine.
the preparation method of sodium sulfobromophthalein is generally to obtain sulfonic acid phenol through sulfonation reaction of phenol, then to obtain sulfonic acid bromophenol through bromination reaction, and finally to obtain sodium sulfobromophthalein through condensation reaction with catechol.
There are some safety information to note when using sodium sulfobromophthalein. It may cause irritation to the skin, eyes and respiratory system when touched, inhaled or ingested. Therefore, it is necessary to wear appropriate protective equipment to avoid direct contact when using sodium sulfobromophthalide. At the same time, keep it in a dry, cool, well-ventilated place, away from fire sources and other combustible substances. In the event of an accident, clean the affected area immediately and seek medical help. Finally, the use of sodium sulfobromide should follow the relevant national and regional regulations and safety procedures.
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: Sulfobromophthalein Sodium Visit Supplier Webpage Request for quotationCAS: 123359-42-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Sulfobromophthalein sodium Request for quotation
CAS: 123359-42-2
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 123359-42-2
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: Sulfobromophthalein sodium Request for quotation
CAS: 123359-42-2
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 123359-42-2
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: Sulfobromophthalein Sodium Visit Supplier Webpage Request for quotation
CAS: 123359-42-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 123359-42-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Sulfobromophthalein Sodium Visit Supplier Webpage Request for quotationCAS: 123359-42-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Sulfobromophthalein sodium Request for quotation
CAS: 123359-42-2
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 123359-42-2
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: Sulfobromophthalein sodium Request for quotation
CAS: 123359-42-2
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 123359-42-2
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: Sulfobromophthalein Sodium Visit Supplier Webpage Request for quotation
CAS: 123359-42-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 123359-42-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



