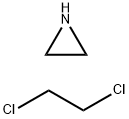
Aziridine,homopolymer,reactionproductswith1,2-dichloroethane
POLYETHYLENEIMINE
CAS: 68130-97-2
Molecular Formula: C4H9Cl2N
Aziridine,homopolymer,reactionproductswith1,2-dichloroethane - Names and Identifiers
Aziridine,homopolymer,reactionproductswith1,2-dichloroethane - Physico-chemical Properties
| Molecular Formula | C4H9Cl2N |
| Molar Mass | 142.02 |
| Density | 1.08g/mLat 25°C |
| Boling Point | 250°C(lit.) |
| Flash Point | >230°F |
| Vapor Presure | 9 mm Hg ( 20 °C) |
| Sensitive | Hygroscopic |
| Refractive Index | n20/D 1.5290 |
Aziridine,homopolymer,reactionproductswith1,2-dichloroethane - Risk and Safety
| WGK Germany | 2 |
| RTECS | TQ7917000 |
| FLUKA BRAND F CODES | 10-34 |
Aziridine,homopolymer,reactionproductswith1,2-dichloroethane - Introduction
The homopolymer of nitrogen propane is a polymer with the chemical formula (C3H6N)n. Its properties depend on the degree of polymerization and the reaction conditions used in the polymerization process. In general, homopolymers of nitropropane are colorless or yellowish solids, have a high molecular weight and a high melting point. It has good heat resistance, chemical stability and mechanical strength at room temperature. In addition, the homopolymer of nitrogen propane also has excellent electrical insulation properties.
Homopolymers of nitrogen propane can be used in many industries. Due to its heat resistance and electrical insulation properties, it is often used as a cable insulation material. In addition, pipes and containers made of nitrogen propane homopolymers are also widely used in the chemical industry.
The preparation of homopolymers of nitrogen propane is usually achieved by polymerization. The reaction generally requires the presence of a polymerization catalyst, such as a metal cyanide. The reaction temperature and time also affect the properties of the polymer.
Regarding the reaction product of 1,2-dichloroethane and nitrogen propane, these two compounds will undergo a substitution reaction called Sn2 reaction under the reaction conditions. This reaction results in the replacement of the chloride ion in 1,2-dichloroethane by the amino group in the nitrogen propane, yielding propylamine chloride (CH3CH2CH2NH2) and ammonium chloride (CH3CH2CH2NH3Cl) as products.
Propylamine Chloride is a colorless liquid with a pungent odor. It can be used as a solvent, surfactant and chemical intermediate. Ammonium chloride is a solid, commonly used as an ion exchanger, water treatment agent and catalyst.
in this reaction, should pay attention to safety issues. 1,2-Dichloroethane is a toxic organic solvent with irritant and carcinogenic effects. Appropriate protective equipment should be worn during operation to ensure a well-ventilated laboratory environment. In addition, care should be taken to avoid contact with other organics and strong oxidants to prevent unsafe reactions from occurring.
Homopolymers of nitrogen propane can be used in many industries. Due to its heat resistance and electrical insulation properties, it is often used as a cable insulation material. In addition, pipes and containers made of nitrogen propane homopolymers are also widely used in the chemical industry.
The preparation of homopolymers of nitrogen propane is usually achieved by polymerization. The reaction generally requires the presence of a polymerization catalyst, such as a metal cyanide. The reaction temperature and time also affect the properties of the polymer.
Regarding the reaction product of 1,2-dichloroethane and nitrogen propane, these two compounds will undergo a substitution reaction called Sn2 reaction under the reaction conditions. This reaction results in the replacement of the chloride ion in 1,2-dichloroethane by the amino group in the nitrogen propane, yielding propylamine chloride (CH3CH2CH2NH2) and ammonium chloride (CH3CH2CH2NH3Cl) as products.
Propylamine Chloride is a colorless liquid with a pungent odor. It can be used as a solvent, surfactant and chemical intermediate. Ammonium chloride is a solid, commonly used as an ion exchanger, water treatment agent and catalyst.
in this reaction, should pay attention to safety issues. 1,2-Dichloroethane is a toxic organic solvent with irritant and carcinogenic effects. Appropriate protective equipment should be worn during operation to ensure a well-ventilated laboratory environment. In addition, care should be taken to avoid contact with other organics and strong oxidants to prevent unsafe reactions from occurring.
Last Update:2024-04-10 22:29:15
Supplier List
Product Name: POLYETHYLENEIMINE Request for quotation
CAS: 68130-97-2
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 68130-97-2
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Product Name: POLYETHYLENEIMINE Request for quotation
CAS: 68130-97-2
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 68130-97-2
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
View History
