90914-41-3
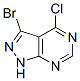
3-Bromo-4-chloro-1H-pyrazolo[3,4-d]pyrimidine
CAS: 90914-41-3
Molecular Formula: C5H2BrClN4
90914-41-3 - Names and Identifiers
| Name | 3-Bromo-4-chloro-1H-pyrazolo[3,4-d]pyrimidine |
| Synonyms | 110922 3-Bromo-4-chloro-1H-pyrazolo[3,4-d]pyrimidine 3-bromo-4-chloro-2H-pyrazolo[3,4-d]pyrimidine 3-BROMO-4-CHLORO-1H-PYRAZOLO[3,4-D]PYRIMIDINE 1H-Pyrazolo[3,4-d]pyriMidine, 3-broMo-4-chloro- 1H-Pyrazolo[3,4-d]pyrimidine, 3-bromo-4-chloro- |
| CAS | 90914-41-3 |
| InChI | InChI=1/C5H2BrClN4/c6-3-2-4(7)8-1-9-5(2)11-10-3/h1H,(H,8,9,10,11) |
90914-41-3 - Physico-chemical Properties
| Molecular Formula | C5H2BrClN4 |
| Molar Mass | 233.45 |
| Density | 2.41±0.1 g/cm3(Predicted) |
| Boling Point | 270.3±50.0 °C(Predicted) |
| Flash Point | 198.988°C |
| Vapor Presure | 0mmHg at 25°C |
| pKa | 8.36±0.20(Predicted) |
| Storage Condition | Keep in dark place,Inert atmosphere,Store in freezer, under -20°C |
| Refractive Index | 1.766 |
90914-41-3 - Risk and Safety
| Hazard Symbols | T - Toxic |
| Risk Codes | 25 - Toxic if swallowed |
| Safety Description | 45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) |
| UN IDs | UN2811 |
| Hazard Class | IRRITANT |
90914-41-3 - Reference Information
| introduction | pyrazolo [3,4-d] pyrimidine derivatives are a class of nitrogen-containing fused ring compounds that have important pharmacological effects due to their structural similarity with purines. related reports also show that they have a variety of biological effects, including anticoagulant, antibacterial and anticancer. |
| preparation | add formamide (150mL) and 10.8g(100.0mmol)3-amino -4-cyanopyrazole 2 into a 500mL three-mouth bottle, heat to 180 ℃ for 10h,TLC monitor the reaction, add water (200mL) to dilute, the reaction system becomes white and turbid. Vacuum suction filtration to obtain white filter cake, wash the filter cake with ice water, vacuum drying to obtain white solid 1H-pyrazolo [3,4-d] pyrimidin-4-amine 13.1g. 6.8g(50.0mmol) 1H-pyrazolo [3,4-d] pyrimidine -4-amine, 16.9g(75.0mmol)N-bromosuccinimide and 60mL N,N-dimethylformamide were added to a 250mL three-mouth bottle, and the reaction system was heated to 100 ℃ for 8h. After TLC monitoring reaction, gray-white crude product was obtained by filtration, and 12.8g of compound was obtained by washing with ice ethanol solution with 98.1% yield. The target compound is prepared by the chlorination reaction of phosphorus oxychloride. |
Last Update:2024-04-09 15:16:37
Supplier List
Product Name: 3-BROMO-4-CHLORO-1H-PYRAZOLO[3,4-D]PYRIMIDINE Request for quotation
CAS: 90914-41-3
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 90914-41-3
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Spot supply
Product Name: 3-Bromo-4-chloro-1H-pyrazolo[3,4-d]pyrimidine Visit Supplier Webpage Request for quotationCAS: 90914-41-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 3-BROMO-4-CHLORO-1H-PYRAZOLO[3,4-D]PYRIMIDINE Request for quotation
CAS: 90914-41-3
Tel: +86-21-56795779
Email: charles7788@worldyachem.com
Mobile: +86-13651600618
QQ: 2850607228
Wechat: 13651600618
WhatsApp: +8613651600618
CAS: 90914-41-3
Tel: +86-21-56795779
Email: charles7788@worldyachem.com
Mobile: +86-13651600618
QQ: 2850607228
Wechat: 13651600618
WhatsApp: +8613651600618
Spot supply
Product Name: 3-BROMO-4-CHLORO-1H-PYRAZOLO[3,4-D]PYRIMIDINE Visit Supplier Webpage Request for quotationCAS: 90914-41-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: 3-BROMO-4-CHLORO-1H-PYRAZOLO[3,4-D]PYRIMIDINE Request for quotation
CAS: 90914-41-3
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 90914-41-3
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Spot supply
Product Name: 3-Bromo-4-chloro-1H-pyrazolo[3,4-d]pyrimidine Visit Supplier Webpage Request for quotationCAS: 90914-41-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 3-BROMO-4-CHLORO-1H-PYRAZOLO[3,4-D]PYRIMIDINE Request for quotation
CAS: 90914-41-3
Tel: +86-21-56795779
Email: charles7788@worldyachem.com
Mobile: +86-13651600618
QQ: 2850607228
Wechat: 13651600618
WhatsApp: +8613651600618
CAS: 90914-41-3
Tel: +86-21-56795779
Email: charles7788@worldyachem.com
Mobile: +86-13651600618
QQ: 2850607228
Wechat: 13651600618
WhatsApp: +8613651600618
Spot supply
Product Name: 3-BROMO-4-CHLORO-1H-PYRAZOLO[3,4-D]PYRIMIDINE Visit Supplier Webpage Request for quotationCAS: 90914-41-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



