89466-08-0
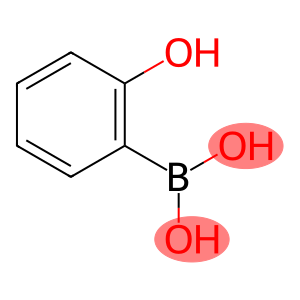
2-Hydroxybenzeneboronic acid
CAS: 89466-08-0
Molecular Formula: C6H7BO3
89466-08-0 - Names and Identifiers
| Name | 2-Hydroxybenzeneboronic acid |
| Synonyms | AKOS BRN-0196 2-Boronophenol RARECHEM AH PB 0223 2-Hydroxyphenylboronic 2-HYDROXYPHENYLBORONIC ACID O-HYDROXYPHENYLBORONIC ACID 2-Hydroxyphenylboronic acid 2-HYDROXYBENZENEBORONIC ACID 2-Hydroxybenzeneboronic acid 2-Boronophenol~2-Hydroxyphenylboronic acid |
| CAS | 89466-08-0 |
| EINECS | 677-277-5 |
| InChI | InChI=1/C6H7BO3/c8-6-4-2-1-3-5(6)7(9)10/h1-4,8-10H |
89466-08-0 - Physico-chemical Properties
| Molecular Formula | C6H7BO3 |
| Molar Mass | 137.93 |
| Density | 1.32±0.1 g/cm3(Predicted) |
| Melting Point | 178-182°C |
| Boling Point | 327.3±44.0 °C(Predicted) |
| Flash Point | 151.7°C |
| Water Solubility | Insoluble in water. |
| Solubility | Acetonitrle (Slightly), DMSO (Slightly) |
| Vapor Presure | 8.28E-05mmHg at 25°C |
| Appearance | Crystalline powder |
| Color | White |
| BRN | 2962938 |
| pKa | 8.61±0.58(Predicted) |
| Storage Condition | Keep in dark place,Sealed in dry,Room Temperature |
| Refractive Index | 1.582 |
| MDL | MFCD01074581 |
89466-08-0 - Risk and Safety
| Risk Codes | R36/37/38 - Irritating to eyes, respiratory system and skin. R41 - Risk of serious damage to eyes R22 - Harmful if swallowed |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. |
| HS Code | 29319000 |
| Hazard Class | IRRITANT |
89466-08-0 - Reference Information
| Introduction | 2-hydroxyphenylboronic acid is an important molecular fragment for the preparation of new drugs for the treatment of diabetes, it is also an important intermediate in organic synthesis of pharmaceuticals and pesticides. It is widely used in Suzuki cross-coupling reaction and is a key intermediate in the preparation of 2-hydroxybiphenyl compounds. Suzuki-Miyaura coupling reaction (SMC) is an important reaction to construct all kinds of C- C single bonds, and its main raw material is organic boric acid compound. |
| Application | 2-hydroxyphenylboronic acid is a boronizing reagent, which can be used as organic synthesis intermediates and pharmaceutical intermediates, mainly used in laboratory research and development process and chemical and pharmaceutical synthesis process. |
Last Update:2024-04-09 20:52:54
Supplier List
Spot supply
Product Name: 2-Hydroxyphenylboronic acid Visit Supplier Webpage Request for quotationCAS: 89466-08-0
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: 2-Hydroxyphenylboronic acid Visit Supplier Webpage Request for quotationCAS: 89466-08-0
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-Hydroxyphenylboronic acid Request for quotation
CAS: 89466-08-0
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 89466-08-0
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Spot supply
Product Name: 2-Hydroxyphenylboronic acid Visit Supplier Webpage Request for quotationCAS: 89466-08-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 2-Hydroxyphenylboronic acid Visit Supplier Webpage Request for quotationCAS: 89466-08-0
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: 2-Hydroxyphenylboronic acid Visit Supplier Webpage Request for quotationCAS: 89466-08-0
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-Hydroxyphenylboronic acid Request for quotation
CAS: 89466-08-0
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 89466-08-0
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Spot supply
Product Name: 2-Hydroxyphenylboronic acid Visit Supplier Webpage Request for quotationCAS: 89466-08-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



