892869-21-5
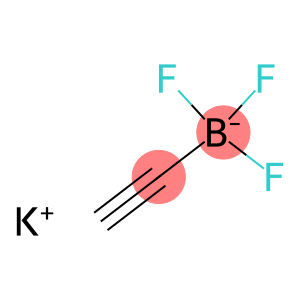
Borate(1-),ethynyltrifluoro-, potassium (1:1), (T-4)-
CAS: 892869-21-5
Molecular Formula: C2HBF3.K
892869-21-5 - Names and Identifiers
| Name | Borate(1-),ethynyltrifluoro-, potassium (1:1), (T-4)- |
| Synonyms | POTASSIUM ethynyltrifluoroborate ETHYNYL(TRIFLUORO)BORANUIDE ethynyl(trifluoro)boranuide Potassium ethynyltrifluoroborate Ethynyltrifluoroborate(1-) potassium Borate(1-),ethynyltrifluoro-, potassium, (T-4)- |
| CAS | 892869-21-5 |
892869-21-5 - Physico-chemical Properties
| Molecular Formula | C2HBF3.K |
| Molar Mass | 131.933 |
| Melting Point | 211.2-212.0 °C (decomp) |
| Storage Condition | under inert gas (nitrogen or Argon) at 2-8°C |
892869-21-5 - Introduction
Boreate (1-),ethynyltrifluoro-, potassium (1:1), (T-4)-is an inorganic compound with the chemical formula C₂ HF-BO₂ K. The following is an introduction to the nature, use, formulation and safety information of Borate(1-),ethynyltrifluoro-, potassium (1:1), (T-4):
Nature:
-Appearance: Borate(1-),ethynyltrifluoro-, potassium (1:1), (T-4)-is a white crystalline solid.
-Melting point: Its melting point is about 200-205°C.
-Solubility: Borate(1-),ethynyltrifluoro-, potassium (1:1), (T-4)-Low solubility in water, soluble in organic solvents such as ethanol, methanol, etc.
Use:
- Borate(1-),ethynyltrifluoro-, potassium (1:1), (T-4)-are commonly used as reagents in organic synthesis, especially as base catalysts.
-It is also used as a catalyst, for example, in organic synthesis reactions, it can promote the synthesis of strongly basic enolate salts.
-In addition, Borate(1-),ethynyltrifluoro-, potassium (1:1), (T-4)-can also be used to catalyze the alternating reaction of halogenated hydrocarbons and borate esters.
Preparation Method:
The preparation of Borate(1-),ethynyltrifluoro-, potassium (1:1), (T-4)-can be completed by the following steps:
1. First, hydrogen fluoride gas is introduced into an organic solvent containing acetylene and trifluoroboric acid to generate fluoroacetylene trifluoroboric acid.
2. then, potassium hydroxide solution is added to the reaction system, and fluoroacetylene trifluoroboric acid is reacted to generate Borate(1-),ethynyltrifluoro-, potassium (1:1), (T-4)-.
3. Finally, the reaction solution was filtered and dried to obtain white crystals of Borate(1-),ethynyltrifluoro-, potassium (1:1), (T-4).
Safety Information:
The specific safety information for Borate(1-),ethynyltrifluoro-, potassium (1:1), (T-4)-is not clear. Due to its strong oxidation, it is necessary to wear gloves, goggles and operate in a well-ventilated place. At the same time, avoid reacting with other chemicals such as hydrogen, ammonia, etc. to avoid danger. If ingested or inhaled, seek medical help immediately.
Nature:
-Appearance: Borate(1-),ethynyltrifluoro-, potassium (1:1), (T-4)-is a white crystalline solid.
-Melting point: Its melting point is about 200-205°C.
-Solubility: Borate(1-),ethynyltrifluoro-, potassium (1:1), (T-4)-Low solubility in water, soluble in organic solvents such as ethanol, methanol, etc.
Use:
- Borate(1-),ethynyltrifluoro-, potassium (1:1), (T-4)-are commonly used as reagents in organic synthesis, especially as base catalysts.
-It is also used as a catalyst, for example, in organic synthesis reactions, it can promote the synthesis of strongly basic enolate salts.
-In addition, Borate(1-),ethynyltrifluoro-, potassium (1:1), (T-4)-can also be used to catalyze the alternating reaction of halogenated hydrocarbons and borate esters.
Preparation Method:
The preparation of Borate(1-),ethynyltrifluoro-, potassium (1:1), (T-4)-can be completed by the following steps:
1. First, hydrogen fluoride gas is introduced into an organic solvent containing acetylene and trifluoroboric acid to generate fluoroacetylene trifluoroboric acid.
2. then, potassium hydroxide solution is added to the reaction system, and fluoroacetylene trifluoroboric acid is reacted to generate Borate(1-),ethynyltrifluoro-, potassium (1:1), (T-4)-.
3. Finally, the reaction solution was filtered and dried to obtain white crystals of Borate(1-),ethynyltrifluoro-, potassium (1:1), (T-4).
Safety Information:
The specific safety information for Borate(1-),ethynyltrifluoro-, potassium (1:1), (T-4)-is not clear. Due to its strong oxidation, it is necessary to wear gloves, goggles and operate in a well-ventilated place. At the same time, avoid reacting with other chemicals such as hydrogen, ammonia, etc. to avoid danger. If ingested or inhaled, seek medical help immediately.
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: Potassium ethynyltrifluoroborate Visit Supplier Webpage Request for quotationCAS: 892869-21-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Potassium ethynyltrifluoroborate Request for quotation
CAS: 892869-21-5
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 892869-21-5
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: Potassium ethynyltrifluoroborate Visit Supplier Webpage Request for quotation
CAS: 892869-21-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 892869-21-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Potassium ethynyltrifluoroborate Visit Supplier Webpage Request for quotationCAS: 892869-21-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Potassium ethynyltrifluoroborate Request for quotation
CAS: 892869-21-5
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 892869-21-5
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: Potassium ethynyltrifluoroborate Visit Supplier Webpage Request for quotation
CAS: 892869-21-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 892869-21-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


