89151-44-0
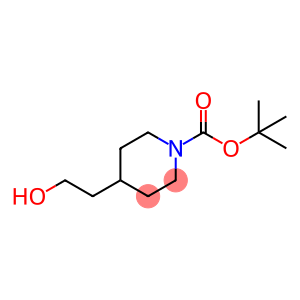
1-Boc-4-(2-hydroxyethyl)piperidine
CAS: 89151-44-0
Molecular Formula: C12H23NO3
89151-44-0 - Names and Identifiers
89151-44-0 - Physico-chemical Properties
| Molecular Formula | C12H23NO3 |
| Molar Mass | 229.32 |
| Density | 1.043 g/mL at 25 °C (lit.) |
| Boling Point | 120-150 °C/0.5 mmHg (lit.) |
| Flash Point | >230°F |
| Vapor Presure | 0mmHg at 25°C |
| Appearance | Brown liquid |
| pKa | 15.10±0.10(Predicted) |
| Storage Condition | Keep in dark place,Sealed in dry,Room Temperature |
| Refractive Index | n20/D 1.4730(lit.) |
| MDL | MFCD03427086 |
| Use | This product is for scientific research only and shall not be used for other purposes. |
89151-44-0 - Risk and Safety
| Hazard Symbols | Xi - Irritant |
| WGK Germany | 3 |
89151-44-0 - Reference Information
| Use | N-Boc-4-piperidine ethanol is an organic intermediate, which can be prepared by the reaction of 4-piperidine ethanol and di-tert-butyl dicarbonate. |
| synthetic method | 4-piperidine ethanol (1.05g,8.13mmol) dissolve in dioxane/water (1: 1,50ml), then add Boc2O(2.13g,9.76mmol) and NaHCO3(8.62g,81.3mmol), the mixture was stirred at room temperature for 24h and then extracted with DCM. The mixed organic phases were washed successively with citric acid (N-Boc-4 solution) and aqueous NaHCO3 solution, followed by drying (Na2SO4), filtering and distilling the solvent to give 1, 4-piperidinethanol (1.77g,95%).. 1HNMR(CDCl3) Φ1.12 (dq,2h, J = 4.6Hz,11.8Hz),1.35-1.55(m,14h),2.70(t,2h, J = 12.8Hz),3.70(t,2h, J = 6.3Hz,-CH2CH2OH),3.95-4.20(m,2H);13C NMR(CDCl3) Δ28.7 (Boc-CH3),32.8,39.5,44.2,60.5,67.3,79.4(-C(CH3)3),155.1(C = O). |
Last Update:2024-04-09 20:52:54
Supplier List
Spot supply
Product Name: 1-Boc-4-(2-hydroxyethyl)piperidine Visit Supplier Webpage Request for quotationCAS: 89151-44-0
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: N-Boc-4-piperidineethanol Visit Supplier Webpage Request for quotationCAS: 89151-44-0
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 1-Boc-4-(2-hydroxyethyl)piperidine Request for quotation
CAS: 89151-44-0
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 89151-44-0
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: N-Boc-4-piperidineethanol Visit Supplier Webpage Request for quotation
CAS: 89151-44-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 89151-44-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 1-Boc-4-(2-hydroxyethyl)piperidine Visit Supplier Webpage Request for quotationCAS: 89151-44-0
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: N-Boc-4-piperidineethanol Visit Supplier Webpage Request for quotationCAS: 89151-44-0
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 1-Boc-4-(2-hydroxyethyl)piperidine Request for quotation
CAS: 89151-44-0
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 89151-44-0
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: N-Boc-4-piperidineethanol Visit Supplier Webpage Request for quotation
CAS: 89151-44-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 89151-44-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



