871125-68-7
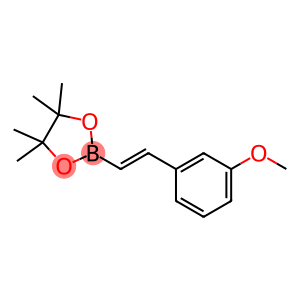
2-[(E)-2-(3-methoxyphenyl)ethenyl]-4,4,5,5-tetramethyl-1,3,2-dioxaborolane
CAS: 871125-68-7
Molecular Formula: C15H21BO3
871125-68-7 - Names and Identifiers
| Name | 2-[(E)-2-(3-methoxyphenyl)ethenyl]-4,4,5,5-tetramethyl-1,3,2-dioxaborolane |
| Synonyms | Trans-2-(3-methoxyphenyl)vinylboronic acidpinacol 3-Methoxy-trans-beta-styrylboronic acid pinacol ester trans-2-(3-Methoxyphenyl)vinylboronic acid pinacol ester (E)-2-(3-Methoxystyryl)-4,4,5,5-tetramethyl-1,3,2-dioxaborolane 2-[(E)-2-(3-methoxyphenyl)ethenyl]-4,4,5,5-tetramethyl-1,3,2-dioxaborolane |
| CAS | 871125-68-7 |
| InChI | InChI=1/C15H21BO3/c1-14(2)15(3,4)19-16(18-14)10-9-12-7-6-8-13(11-12)17-5/h6-11H,1-5H3/b10-9+ |
871125-68-7 - Physico-chemical Properties
| Molecular Formula | C15H21BO3 |
| Molar Mass | 260.14 |
| Density | 1.02±0.1 g/cm3(Predicted) |
| Melting Point | 36-42°C(lit.) |
| Boling Point | 313.5±44.0 °C(Predicted) |
| Flash Point | 110°C |
| Vapor Presure | 0.000908mmHg at 25°C |
| Storage Condition | 2-8°C |
| Refractive Index | 1.503 |
871125-68-7 - Risk and Safety
| WGK Germany | 3 |
871125-68-7 - Introduction
2-[(E)-2-(3-methoxyphenyl)ethenyl]-4,4, 5,5-tetrathyl-1, 3,2-dioxaborolane is an organic compound with the chemical formula C14H19BO2. The following is a description of the properties, uses, preparation and safety information of the compound:
Nature:
-Appearance: 2-[(E)-2-(3-methoxygenyl) ethenyl]-4,4,5,5-tetramethyl-1,3,2-dioxaborolane is a colorless to pale yellow oily liquid.
-Melting point and boiling point: The melting point and boiling point information of this compound is not available.
-Solubility: 2-[(E)-2-(3-methoxyyl) ethenyl]-4,4,5,5-tetramethyl-1,3,2-dioxaborolane can be dissolved in organic solvents such as ethanol, dimethyl sulfoxide and dichloromethane.
Use:
- 2-[(E)-2-(3-methoxyphenyl)ethenyl]-4,4, 5,5-tetrathyl-1, 3,2-dioxaborolane is an important intermediate in organic synthesis. It is often used in organic synthesis reactions, such as Suzuki-Miyaura coupling reactions, as an important reagent for the construction of compound bonds.
-The compounds can also be used for the preparation of other pharmaceuticals, chemicals and heterocyclic compounds.
Preparation Method: The preparation method of
- 2-[(E)-2-(3-methoxyphenyl)ethenyl]-4,4,5,5-tetramethyl-1,3,2-dioxaborolane is more complicated, and the common method is obtained by chemical synthesis reaction. The preparation method involves the corresponding starting materials and reagents, and the specific synthetic route needs to be carried out in a laboratory environment.
Safety Information:
- 2-[(E)-2-(3-methoxyphenyl)ethenyl]-4,4,5,5-tetramethyl-1,3,2-dioxaborolane is generally stable under normal processing conditions, but may decompose or react under high temperature, high pressure or in the presence of organic solvents.
-Avoid skin contact and avoid inhalation or ingestion during use. Wear appropriate protective gloves, glasses and face mask during operation. When handling chemicals, it is important to follow safe operating procedures and properly dispose of waste materials.
Nature:
-Appearance: 2-[(E)-2-(3-methoxygenyl) ethenyl]-4,4,5,5-tetramethyl-1,3,2-dioxaborolane is a colorless to pale yellow oily liquid.
-Melting point and boiling point: The melting point and boiling point information of this compound is not available.
-Solubility: 2-[(E)-2-(3-methoxyyl) ethenyl]-4,4,5,5-tetramethyl-1,3,2-dioxaborolane can be dissolved in organic solvents such as ethanol, dimethyl sulfoxide and dichloromethane.
Use:
- 2-[(E)-2-(3-methoxyphenyl)ethenyl]-4,4, 5,5-tetrathyl-1, 3,2-dioxaborolane is an important intermediate in organic synthesis. It is often used in organic synthesis reactions, such as Suzuki-Miyaura coupling reactions, as an important reagent for the construction of compound bonds.
-The compounds can also be used for the preparation of other pharmaceuticals, chemicals and heterocyclic compounds.
Preparation Method: The preparation method of
- 2-[(E)-2-(3-methoxyphenyl)ethenyl]-4,4,5,5-tetramethyl-1,3,2-dioxaborolane is more complicated, and the common method is obtained by chemical synthesis reaction. The preparation method involves the corresponding starting materials and reagents, and the specific synthetic route needs to be carried out in a laboratory environment.
Safety Information:
- 2-[(E)-2-(3-methoxyphenyl)ethenyl]-4,4,5,5-tetramethyl-1,3,2-dioxaborolane is generally stable under normal processing conditions, but may decompose or react under high temperature, high pressure or in the presence of organic solvents.
-Avoid skin contact and avoid inhalation or ingestion during use. Wear appropriate protective gloves, glasses and face mask during operation. When handling chemicals, it is important to follow safe operating procedures and properly dispose of waste materials.
Last Update:2024-04-09 02:00:48
Supplier List
Spot supply
Product Name: trans-2-(3-Methoxyphenyl)vinylboronic acid pinacol ester Visit Supplier Webpage Request for quotationCAS: 871125-68-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: TRANS-2-(3-METHOXYPHENYL)VINYLBORONIC ACID PINACOL ESTER, 96% Request for quotation
CAS: 871125-68-7
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 871125-68-7
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: TRANS-2-(3-METHOXYPHENYL)VINYLBORONIC ACID PINACOL ESTER Visit Supplier Webpage Request for quotation
CAS: 871125-68-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 871125-68-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: trans-2-(3-Methoxyphenyl)vinylboronic acid pinacol ester Visit Supplier Webpage Request for quotationCAS: 871125-68-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: TRANS-2-(3-METHOXYPHENYL)VINYLBORONIC ACID PINACOL ESTER, 96% Request for quotation
CAS: 871125-68-7
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 871125-68-7
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: TRANS-2-(3-METHOXYPHENYL)VINYLBORONIC ACID PINACOL ESTER Visit Supplier Webpage Request for quotation
CAS: 871125-68-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 871125-68-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


