811803-05-1
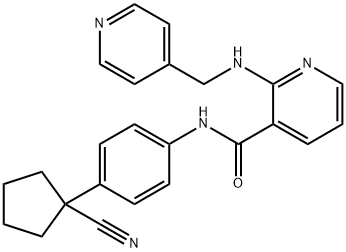
N-[4-(1-Cyanocyclopentyl)phenyl]-2-[(4-pyridinylmethyl)amino]nicotinamide methanesulfonate (1:1)
CAS: 811803-05-1
Molecular Formula: C24H23N5O
811803-05-1 - Names and Identifiers
| Name | N-[4-(1-Cyanocyclopentyl)phenyl]-2-[(4-pyridinylmethyl)amino]nicotinamide methanesulfonate (1:1) |
| Synonyms | Aptinib CS-1160 Apatinib Mesylate APA Apatinib Mesylate Apatinib (YN968D1) APATINIB FREE BASE (YN968D1 FREE BASE) N-(4-(1-cyanocyclopentyl)phenyl)-2-(pyridin-4-ylMethylaMino)nicotinaMide |
| CAS | 811803-05-1 |
| EINECS | 1592732-453-0 |
| InChI | InChI=1S/C24H23N5O.CH4O3S/c25-17-24(11-1-2-12-24)19-5-7-20(8-6-19)29-23(30)21-4-3-13-27-22(21)28-16-18-9-14-26-15-10-18;1-5(2,3)4/h3-10,13-15H,1-2,11-12,16H2,(H,27,28)(H,29,30);1H3,(H,2,3,4) |
811803-05-1 - Physico-chemical Properties
| Molecular Formula | C24H23N5O |
| Molar Mass | 397.47 |
| Density | 1.27 |
| Boling Point | 578.2±50.0 °C(Predicted) |
| pKa | 11.93±0.70(Predicted) |
| Physical and Chemical Properties |
|
811803-05-1 - Reference Information
| drugs for advanced gastric cancer | apatinib it is the first small molecule anti-angiogenic targeted drug confirmed to be safe and effective in advanced gastric cancer in the world. It is also the single drug with the best efficacy after the failure of standardized treatment for advanced gastric cancer. In addition, Apatinib is also the only oral drug in the treatment of gastric cancer, which will greatly improve the Recovery Rate of patients. In June of 2014, the clinical study of the drug was selected as a report by the American Society of Clinical Oncology (ASCO), which is the first time that China's innovative drug research has presented a report at the world's top academic conference, this is the first time to be selected for excellent research in this annual meeting. on December 13, 2014, Jiangsu Hengrui Pharmaceutical Co., Ltd. announced that Apatinib, a new drug approved by the State Food and Drug Administration for the treatment of advanced gastric cancer. Officially listed in China. Apatinib, Apatinib mesylate tablets, is the first confirmed safe and effective oral small-molecule anti-angiogenic targeted drug in the world after failure of standard chemotherapy for advanced gastric cancer, it can significantly prolong the overall survival of patients with advanced gastric cancer and bring new hope for the treatment of patients with advanced gastric cancer. (2015-09-23) |
| pharmacological action | Apatinib is a small molecule targeted drug with anti-VEGFR-2, for the population of patients with advanced gastric cancer who have failed second-line chemotherapy, by highly selective competition for intracellular receptor -2 ATP binding sites, blocking downstream signal transduction, to inhibit the formation of tyrosine kinases to inhibit tumor angiogenesis, and ultimately achieve the purpose of treatment of tumors. The clinical trial was jointly led by Professor Qin Shuyi and Professor Li Jin from Nanjing Bayi Hospital of PLA, and received strong support from 38 hospitals across the country. A total of 273 patients were enrolled: 1 randomly assigned to oral Apatinib or placebo treatment. Over a one-and-a-half year study period, Apatinib significantly prolonged overall survival (OS) by 55 days compared with placebo (195 days vs.140 days, P<0.016). |
| Apatinib Mesylate | Apatinib Mesylate (Apatinib Mesylate) is a small molecule tyrosine kinase inhibitor independently developed by Jiangsu Hengrui Pharmaceutical Co., Ltd., by selectively inhibiting the tyrosine kinase activity of vascular endothelial growth factor receptor 2(VEGFR-2), thereby inhibiting tumor angiogenesis, inhibit tumor growth. It is intended to be used in the treatment of advanced gastric cancer with failure of second-line chemotherapy ". Dosage forms for tablets, specifications: 250mg, 375mg, 425mg. Apatinib is a new generation tyrosine kinase inhibitor that blocks tumor angiogenesis. Apatinib is the world's first confirmed to be able to treat advanced gastric cancer safe and effective small molecule anti-angiogenesis targeted drugs, but also advanced gastric cancer after standard chemotherapy failure, can significantly prolong the survival of the single drug. At the same time, the drug is the only oral drug for gastric cancer, which can effectively improve the compliance of patients and significantly reduce the cost of treatment. The mechanism of action of Apatinib is: highly selective competition for VEGFR-2 of the ATP binding sites in cells, blocking downstream signal transduction, inhibiting tumor angiogenesis, thereby treating tumor, can significantly prolong the survival of patients with advanced gastric cancer. Figure 1 shows the Apatinib (Apatinib mesylate tablets) |
| market prospects | gastric cancer is a high-incidence cancer in China, with a high incidence Mortality Rate. According to relevant statistics, there were 952000 new cases of harm in the world in 2013, of which China accounted for 47%, and it occupied the second place among malignant tumors in China Incidence Rate. About 325300 people die from harm every year, it ranks third in the Mortality Rate malignant tumors in China. Because the early symptoms of cancer are not obvious and the routine gastroscopy is not universal, about 70% of the patients are diagnosed with advanced gastric cancer at the time of treatment. The existing treatment is effective and the prognosis is poor, its five-year survival rate does not exceed 20%. In recent decades, although a lot of manpower and material resources have been invested, the research and development of new drugs for advanced gastric cancer after the failure of standardized treatment has not made a breakthrough, resulting in the lack of effective treatment for this group of people, the burden on society and the state is enormous. Apatinib is a new small molecule targeted drug, which can significantly prolong the survival time of patients with advanced gastric cancer and greatly reduce the cost of patients by inhibiting tumor angiogenesis. |
| side effects | , the Apatinib all clinical studies safety database included 36 healthy subjects who took 1 dose and 1230 patients with solid tumors who took at least 1 dose. In Phase I, II, and III trials, 229 subjects received a dose of 850mg qd (6 in phase I trials, 47 in Phase II trials, and 176 in Phase III trials). The types of adverse reactions observed are similar to those of other antiangiogenic agents that have become major targets. Common adverse reactions (≥ 5%) included hematologic toxicity (leukopenia, granulocytopenia, thrombocytopenia, decreased hemoglobin) and proteinuria, hypertension, hand-foot syndrome, gastrointestinal reactions (Diarrhea, abdominal Pain, Nausea, Vomit, loss of appetite), Fatigue, back pain, hoarseness. Major laboratory abnormalities included abnormal liver enzymes (elevated transaminases, elevated bilirubin, elevated alkaline phosphatase, elevated gamma-glutamyl transpeptidase, elevated lactate dehydrogenase), abnormal electrolytes (low potassium, low calcium, low sodium). The most common serious adverse reaction was upper gastrointestinal bleeding. Most treatment-related adverse reactions are of grade 1-2 severity and can be managed by temporary discontinuation of Apatinib mesylate, dose reduction, or symptomatic treatment. There were no unexpected toxicities, similar to the Phase III reports. |
Last Update:2024-04-09 02:00:12
Supplier List
Spot supply
Product Name: Apatinib Visit Supplier Webpage Request for quotationCAS: 811803-05-1
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: Apatinib Visit Supplier Webpage Request for quotationCAS: 811803-05-1
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Spot supply
Product Name: Apatinib Visit Supplier Webpage Request for quotationCAS: 811803-05-1
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: Apatinib Visit Supplier Webpage Request for quotationCAS: 811803-05-1
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
View History

