81098-60-4
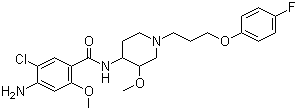
Cisapride
CAS: 81098-60-4
Molecular Formula: C23H29ClFN3O4
81098-60-4 - Names and Identifiers
81098-60-4 - Physico-chemical Properties
| Molecular Formula | C23H29ClFN3O4 |
| Molar Mass | 465.95 |
| Density | 1.29±0.1 g/cm3(Predicted) |
| Melting Point | 109.8 °C |
| Boling Point | 605.4±55.0 °C(Predicted) |
| Water Solubility | 9.319mg/L(30 ºC) |
| Appearance | Form solid, color white |
| pKa | 13.01±0.40(Predicted) |
| Storage Condition | Keep in dark place,Inert atmosphere,Room temperature |
| Physical and Chemical Properties | Chemical Properties Monohydrate: Crystallized from 2-propanol, melting point 109.8 ℃. () type: melting point 129 ℃. Type (-): melting point 125 ℃. Type (): melting point 131~133 ℃. |
| Use | Use of gastrointestinal motility drugs, can strengthen and coordinate gastrointestinal movement, prevent food retention and reflux, and thus have antiemetic effect. There is also a strong stabilizing effect, which can be used as a treatment for mental illness. There is no blockade of dopamine receptors and direct stimulation of cholinergic receptors, so there are no corresponding side effects. Used for gastroparesis, gastroesophageal reflux, upper gastrointestinal discomfort, chronic constipation, pseudo intestinal obstruction, etc. |
81098-60-4 - Risk and Safety
| Hazard Symbols | Xi - Irritant |
| Risk Codes | 41 - Risk of serious damage to eyes |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S39 - Wear eye / face protection. |
| WGK Germany | 2 |
| RTECS | CU9372000 |
81098-60-4 - Upstream Downstream Industry
81098-60-4 - Preparation solution concentration reference
| 1mg | 5mg | 10mg | |
|---|---|---|---|
| 1 mM | 2.146 ml | 10.731 ml | 21.462 ml |
| 5 mM | 0.429 ml | 2.146 ml | 4.292 ml |
| 10 mM | 0.215 ml | 1.073 ml | 2.146 ml |
| 5 mM | 0.043 ml | 0.215 ml | 0.429 ml |
81098-60-4 - Uses and synthesis methods
Pharmacological action
This product is a total gastrointestinal motility agent. It can strengthen and coordinate gastrointestinal movement to prevent food retention and reflux. Its mechanism of action is mainly to enhance gastrointestinal motility by selectively promoting the release of acetylcholine in the posterior nerve of the enteric myenteric plexus. Animal experiments have confirmed that this product can accelerate gastric peristalsis, enhance the digestive activity of the antrum-duodenum, coordinate the movement of the antrum-duodenum, small intestine and large intestine, and shorten the intestinal movement time. In the human body, this product can enhance esophageal peristalsis, increase the tension of the lower esophageal sphincter, prevent gastric contents from flowing back into the esophagus and improve the clearance rate of the esophagus; it can strengthen the contraction of the stomach and the twelve-finged intestine, and improve the gastric antrum-The coordination function of the twelve-finged intestine prevents the twelve-finged intestine-gastric reflux, promotes the emptying of the stomach and twelve-finged intestine; can promote the peristalsis of the small intestine and large intestine.
this product does not affect the gastrointestinal submucosal nerve plexus, so it does not change the secretion of gastrointestinal mucosa. At the same time, because this product does not inhibit the activity of acetylcholinesterase and has no dopamine receptor blocking effect, it does not increase gastric acid secretion, nor does it affect the level of plasma prolactin, and basically has no central inhibitory effect.
Pharmacokinetics
oral administration absorbs rapidly, the blood drug concentration peaks at 1~2h, the protein binding rate is 98%, the bioavailability is 40% ~ 50%, and the half-life is 7~10h. It is metabolized as norcisapride in the antibody, and the 90% is excreted through urine and feces, and a small amount is excreted by milk.
indication
1. Mainly used for functional dyspepsia, relieve upper abdominal fullness, early satiety, nausea, vomiting, belching, upper abdominal burning and other symptoms. This product can also be used to treat chronic, excessive nausea and vomiting in young children who cannot be controlled by posture and diet measures.
2. For the treatment and maintenance of gastroesophageal reflux diseases, including esophagitis.
3. For gastroparesis caused by nerve injury, vagotomy, and partial gastrectomy.
4. It can restore the propulsive movement of the colon and is used for the long-term treatment of chronic constipation.
5. It can be used for propulsive peristalsis deficiency and gastrointestinal content retention caused by intestinal pseudo-obstruction related to motor dysfunction.
adverse reactions
1. a few patients can have transient abdominal cramps, abdominal ringing and diarrhea, and the reduction can disappear.
2. Reports of occasional allergic reactions (eg, rash, urticaria, pruritus, bronchospasm), mild transient headache or dizziness, and dose-related frequent urination.
3. Rare: ① There are very few reports of arrhythmia (including ventricular tachycardia, ventricular fibrillation, torsade de pointes, QT interval prolongation). Most patients often take other drugs (including CYP 3A4 enzyme inhibitor), or have heart disease, or have risk factors for arrhythmia. ② Reports of rare reversible liver dysfunction (possibly with cholestasis). ③ There are also reports of male mammary gland development and galacto (the relationship with this product is not clear). According to individual reports, this product can affect the central nervous system, leading to epilepsy, extrapyramidal reactions, etc.
Taboo
1. people who are allergic to this drug.
2. Those with bradycardia.
3. Patients with prolonged QT interval (including congenital prolonged QT interval) or a family history of congenital prolonged QT interval syndrome.
4. Patients with lung, liver and kidney insufficiency.
5. Infants and young children.
precautions
1. Children who still cannot control galactorrhea (nausea) and vomiting on body position and dietary measures can take this product suspension.
2. When the dosage of drugs related to this product needs to be determined, the blood concentration of these drugs should be monitored.
3. If syncope, increased heart rate or arrhythmia occur during medication, or QT interval of ECG exceeds 0.45 seconds, this product should be stopped immediately.
4. If abdominal cramps occur after medication, the dose can be halved.
5. The following conditions should be used with caution: ① Patients with increased gastrointestinal function can cause risks. ② Those with a family history of sudden death.
6.FDA's pregnancy safety rating for this drug is C.
Production method
3-methoxy-4-aminopiperidine and 4-acetamido-2-methoxy-5-chlorobenzoic acid are dehydrated to generate 4-acetamido-2-methoxy-5-chloro-N-(3-methoxy-4-piperidinyl) benzamide, and then alkylation with p-flufenyl propyl ether on nitrogen, acidification and hydrolysis to generate cisapride.
Supplier List
CAS: 81098-60-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 81098-60-4
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 81098-60-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 81098-60-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 81098-60-4
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 81098-60-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025


