7657-09-2
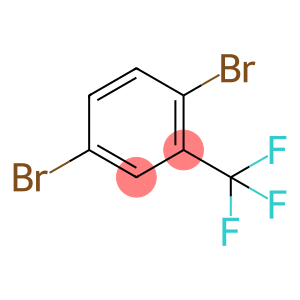
2,5-dibromobenzotrifluoride
CAS: 7657-09-2
Molecular Formula: C7H3Br2F3
7657-09-2 - Names and Identifiers
7657-09-2 - Physico-chemical Properties
| Molecular Formula | C7H3Br2F3 |
| Molar Mass | 303.9 |
| Density | 1.956g/cm3 |
| Melting Point | 49-51°C(lit.) |
| Boling Point | 47 °C |
| Flash Point | >230°F |
| Solubility | soluble in Methanol |
| Vapor Presure | 0.282mmHg at 25°C |
| Appearance | Solid |
| Color | White to Light yellow |
| BRN | 2261093 |
| Storage Condition | Sealed in dry,Room Temperature |
| Refractive Index | 1.52 |
| MDL | MFCD00013554 |
7657-09-2 - Risk and Safety
| Hazard Symbols | Xi - Irritant |
| Risk Codes | 36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. |
| UN IDs | 3276 |
| WGK Germany | 3 |
| Hazard Class | IRRITANT |
7657-09-2 - Reference Information
| uses | bromobenzene compounds are important organic synthesis intermediates, which are widely used in medicine, synthesis of functional chemicals such as pesticides. |
| preparation | preparation of 2, 5-dibromotrifluorobenzene benzene trifluoride was prepared by bromination reaction using benzene trifluoride as starting material, the synthesis reaction formula is shown in the figure below: Fig. 1 2, the commonly used bromination reagents in the synthesis reaction formula of 5-dibromotrifluorobenzene mainly include: elemental bromine, organic bromination reagent, perbromo type quaternary ammonium salt and Quaternary phosphorus salt, tetrabromocyclic ketone, perbrominated pyridinium hydrobromide, N-bromosuccinimide, dibromohydantoin, etc. This time a new synthetic brominating agent is used, which is a 2: 1: 1 type complex of N,N-dimethylacetamide, hydrobromic acid, Pure bromine. The brominating agent is a mild and highly selective brominating agent, and when it is reacted with aryl acetone, bromine can selectively replace the hydrogen atom in the alpha position, and the reaction can stay in the mono-bromination stage. The process conditions of bromination reaction were studied. Experimental operation N,N-dimethylacetamide and 40% hydrobromic acid solution were added to the reaction flask, and the mixture was stirred. Bromine was added dropwise at room temperature, and the mixture was further stirred and filtered to obtain a solid bis (dimethylacetamido) trihydrogen bromide. A predetermined amount of BIS (dimethylacetamido) hydrogen tribromide, benzene trifluoride and 60ml of methanol were added to a 250ml three-necked flask equipped with a thermometer and a condenser tube, and the mixture was stirred and reacted at room temperature for 2 hours. The residue was dissolved by adding chloroform, washed with water, and the organic layer was dried overnight by adding anhydrous sodium sulfate. The solvent was removed under reduced pressure and purified by silica gel column chromatography to obtain 2, 5-dibromotrifluorobenzene. |
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: 2,5-Dibromobenzotrifluoride Visit Supplier Webpage Request for quotationCAS: 7657-09-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2,5-DIBROMOBENZOTRIFLUORIDE Request for quotation
CAS: 7657-09-2
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 7657-09-2
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: 2,5-Dibromobenzotrifluoride Visit Supplier Webpage Request for quotation
CAS: 7657-09-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 7657-09-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 2,5-Dibromobenzotrifluoride Visit Supplier Webpage Request for quotationCAS: 7657-09-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2,5-DIBROMOBENZOTRIFLUORIDE Request for quotation
CAS: 7657-09-2
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 7657-09-2
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: 2,5-Dibromobenzotrifluoride Visit Supplier Webpage Request for quotation
CAS: 7657-09-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 7657-09-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


