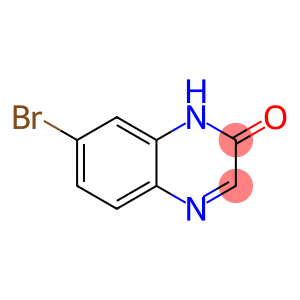
7-broMo-1,2-dihydroquinoxalin-2-one
7-Bromo-2(1H)-quinoxalinone
CAS: 82031-32-1
Molecular Formula: C8H5BrN2O
7-broMo-1,2-dihydroquinoxalin-2-one - Names and Identifiers
| Name | 7-Bromo-2(1H)-quinoxalinone |
| Synonyms | 7-bromoquinoxalin-2-ol 7-Bromoquinoxalin-2(1H) 2-Quinoxalinol, 7-bromo- 7-Bromoquinoxalin-2(1H)-one 7-bromoquinoxalin-2(1H)-one 7-Bromo-2(1H)-quinoxalinone 7-Bromo-1H-quinoxalin-2-one 2(1H)-quinoxalinone, 7-bromo- 2(1H)-Quinoxalinone, 7-bromo- 7-broMo-1,2-dihydroquinoxalin-2-one |
| CAS | 82031-32-1 |
| InChI | InChI=1/C8H5BrN2O/c9-5-1-2-6-7(3-5)11-8(12)4-10-6/h1-4H,(H,11,12) |
7-broMo-1,2-dihydroquinoxalin-2-one - Physico-chemical Properties
| Molecular Formula | C8H5BrN2O |
| Molar Mass | 225.04 |
| Density | 1.82±0.1 g/cm3(Predicted) |
| pKa | 8.23±0.70(Predicted) |
| Storage Condition | Sealed in dry,Room Temperature |
| Refractive Index | 1.72 |
7-broMo-1,2-dihydroquinoxalin-2-one - Reference Information
| Overview | 7-bromo-2 (1H)-quinoxalinone is a commonly used quinolone pharmaceutical intermediate, the quinolone intermediate can be prepared from O-Phenylenediamine and ethylene glycol, and obtained by bromination. |
| Application | 7-bromo-2 (1H)-quinoxalinone is an intermediate in organic synthesis and a pharmaceutical intermediate, can be used in laboratory research and development process and chemical and pharmaceutical synthesis process. |
| preparation | O-phenylenediamine (200g,1.85 mol) was dissolved in ethanol (1.2 L). Ethylene glycol (50% in toluene, 450ml) was added dropwise. The reaction was heated to 50 °c for 12 hours and then cooled to 5 °c for 1 hour. The mixture was filtered and the solid was washed with water in 92% yield calculated as O-phenylenediamine. The quinolone (250g,1.7 mol) was dissolved in acetic acid (4500 mL). A mixture of acetic acid (988 mL) and bromine (108 mL,2.1 mol) was added dropwise and the mixture was stirred at room temperature for 12 hours and then heated to 60 °c for 12 hours. After cooling to room temperature, the reaction was filtered and the solid was washed with water. The wet cake (500g) was then dissolved in 85% ml of methanol and heated to 60 °c, then filtered and dried at 60 °c to give 7-bromo-2 (1H) in yield.-Quinoxalinone. |
Last Update:2024-04-09 19:05:15
Supplier List
Spot supply
Product Name: 7-Bromoquinoxalin-2(1H)-one Visit Supplier Webpage Request for quotationCAS: 82031-32-1
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 7-bromoquinoxalin-2(1H)-one Request for quotation
CAS: 82031-32-1
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 82031-32-1
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: 7-bromoquinoxalin-2(1H)-one Visit Supplier Webpage Request for quotation
CAS: 82031-32-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 82031-32-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 7-Bromoquinoxalin-2(1H)-one Visit Supplier Webpage Request for quotationCAS: 82031-32-1
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 7-bromoquinoxalin-2(1H)-one Request for quotation
CAS: 82031-32-1
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 82031-32-1
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: 7-bromoquinoxalin-2(1H)-one Visit Supplier Webpage Request for quotation
CAS: 82031-32-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 82031-32-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


