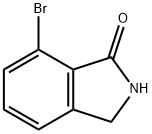
7-Bromoisoindolinone
7-bromoisoindolin-1-one
CAS: 200049-46-3
Molecular Formula: C8H6BrNO
7-Bromoisoindolinone - Names and Identifiers
| Name | 7-bromoisoindolin-1-one |
| Synonyms | 7-Bromoisoindolinone 7-Bromo-1-isoindolinone 7-bromoisoindolin-1-one 7-Bromo-2,3-dihydro-isoindol-1-one 7-bromo-2,3-dihydro-isoindole-1-one 7-bromo-2,3-dihydro-1H-isoindol-1-one 7-broMo-2,3-dihydro-1H-isoindol-1-one 1H-Isoindol-1-one, 7-broMo-2,3-dihydro- 1H-Isoindol-1-one, 7-bromo-2,3-dihydro- |
| CAS | 200049-46-3 |
| InChI | InChI=1/C8H6BrNO/c9-6-3-1-2-5-4-10-8(11)7(5)6/h1-3H,4H2,(H,10,11) |
7-Bromoisoindolinone - Physico-chemical Properties
| Molecular Formula | C8H6BrNO |
| Molar Mass | 212.04 |
| Density | 1.666±0.06 g/cm3(Predicted) |
| Boling Point | 452.2±45.0 °C(Predicted) |
| Flash Point | 227.266°C |
| Vapor Presure | 0mmHg at 25°C |
| pKa | 13.36±0.20(Predicted) |
| Storage Condition | Sealed in dry,Room Temperature |
| Refractive Index | 1.625 |
7-Bromoisoindolinone - Reference Information
| Uses | 7-bromo-2, 3-dihydro-isoindole-1-one is a useful research chemical for organic synthesis and other chemical processes. |
| Preparation | The condensation reaction of o-halomethyl or o-acyl benzoate with amines is a traditional method for synthesizing iso-indole ketone compounds. The selective reduction of phthalimide and various organic transformations of benzo [c] pyrrolidone derivatives are also used to synthesize iso-indole ketone derivatives. In this paper, the target compound 7-bromo-2, 3-dihydro-isoindole-1-one was prepared by ring-closing reaction with methyl 2-bromomethyl-3-chlorobenzoate and liquid ammonia as starting materials. The synthesis reaction formula is as follows: Experimental operation: Methyl 2-bromomethyl-3-chlorobenzoate and liquid ammonia are reacted in 20 mL methanol solution at 60°C under stirring to the end point (TLC detection). Chilled to room temperature, extracted with ethyl acetate, the organic phase was washed with saturated salt water for 3 times, anhydrous sodium sulfate was dried, concentrated and evaporated, and the residue was purified by silica gel column chromatography (eluent: ethyl acetate/petroleum ether, V/ V = 1/5) to obtain compound 7-bromo-2, 3-dihydro-indole-1-one. |
Last Update:2024-04-09 02:00:01
Supplier List
Spot supply
Product Name: 7-Bromo-2,3-dihydro-isoindol-1-one Visit Supplier Webpage Request for quotationCAS: 200049-46-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 7-Bromo-2,3-dihydro-isoindol-1-one Request for quotation
CAS: 200049-46-3
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 200049-46-3
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Product Name: 7-Bromo-2,3-dihydro-isoindol-1-one Visit Supplier Webpage Request for quotation
CAS: 200049-46-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 200049-46-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 7-Bromo-2,3-dihydro-isoindol-1-one Visit Supplier Webpage Request for quotationCAS: 200049-46-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 7-Bromo-2,3-dihydro-isoindol-1-one Request for quotation
CAS: 200049-46-3
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 200049-46-3
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Product Name: 7-Bromo-2,3-dihydro-isoindol-1-one Visit Supplier Webpage Request for quotation
CAS: 200049-46-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 200049-46-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


