634151-25-0
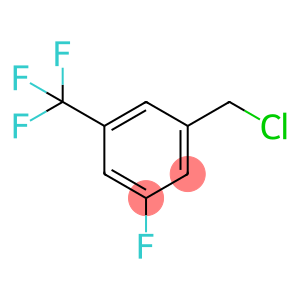
3-Fluoro-5-trifluoromethylbenzyl chloride
CAS: 634151-25-0
Molecular Formula: C8H5ClF4
634151-25-0 - Names and Identifiers
634151-25-0 - Physico-chemical Properties
| Molecular Formula | C8H5ClF4 |
| Molar Mass | 212.57 |
| Density | 1.372g/cm3 |
| Boling Point | 171.2°C at 760 mmHg |
| Flash Point | 67.2°C |
| Vapor Presure | 1.89mmHg at 25°C |
| Storage Condition | 2-8°C |
| Refractive Index | 1.442 |
634151-25-0 - Introduction
chloride is an organic compound with the chemical formula of C8H5ClF4, and its molecules contain elements such as fluorine, chlorine and carbon.
Nature:
chloride is a colorless to yellowish liquid with a pungent odor. It is soluble in organic solvents such as diethyl ether and dichloromethane at room temperature, but hardly soluble in water. It has a low boiling point and a high melting point. It should be noted that vapors of the compound may be irritating to the eyes, skin and respiratory tract.
Use:
chloride can be used as an important starting material in organic synthesis for the preparation of other organic compounds. It can be used as an intermediate in the synthesis of pesticides, and can also be used in the fields of drugs, dyes and fragrances.
Preparation Method:
The preparation of calcium chloride is usually obtained by reacting 3-fluoro-5-trifluoromethylbenzyl alcohol with sodium hypochlorite. The specific procedure involves reacting 3-fluoro-5-trifluoromethylbenzyl alcohol with sodium hypochlorite in a suitable solvent and acidifying to obtain the final product.
Safety Information:
chloride is a hazardous chemical and care should be taken to avoid skin contact, eye contact and inhalation of its vapors. Wear protective gloves, goggles and face shields when in use. If skin contact occurs, rinse immediately with plenty of water. If accidentally inhaled or taken by mistake, should immediately go to the hospital for treatment.
Nature:
chloride is a colorless to yellowish liquid with a pungent odor. It is soluble in organic solvents such as diethyl ether and dichloromethane at room temperature, but hardly soluble in water. It has a low boiling point and a high melting point. It should be noted that vapors of the compound may be irritating to the eyes, skin and respiratory tract.
Use:
chloride can be used as an important starting material in organic synthesis for the preparation of other organic compounds. It can be used as an intermediate in the synthesis of pesticides, and can also be used in the fields of drugs, dyes and fragrances.
Preparation Method:
The preparation of calcium chloride is usually obtained by reacting 3-fluoro-5-trifluoromethylbenzyl alcohol with sodium hypochlorite. The specific procedure involves reacting 3-fluoro-5-trifluoromethylbenzyl alcohol with sodium hypochlorite in a suitable solvent and acidifying to obtain the final product.
Safety Information:
chloride is a hazardous chemical and care should be taken to avoid skin contact, eye contact and inhalation of its vapors. Wear protective gloves, goggles and face shields when in use. If skin contact occurs, rinse immediately with plenty of water. If accidentally inhaled or taken by mistake, should immediately go to the hospital for treatment.
Last Update:2024-04-09 02:00:47
Supplier List
Product Name: 3-FLUORO-5-TRIFLUOROMETHYLBENZYL CHLORIDE Request for quotation
CAS: 634151-25-0
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 634151-25-0
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Product Name: 3-FLUORO-5-TRIFLUOROMETHYLBENZYL CHLORIDE Visit Supplier Webpage Request for quotation
CAS: 634151-25-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 634151-25-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: 3-FLUORO-5-TRIFLUOROMETHYLBENZYL CHLORIDE Request for quotation
CAS: 634151-25-0
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 634151-25-0
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Product Name: 3-FLUORO-5-TRIFLUOROMETHYLBENZYL CHLORIDE Visit Supplier Webpage Request for quotation
CAS: 634151-25-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 634151-25-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History

