621-54-5
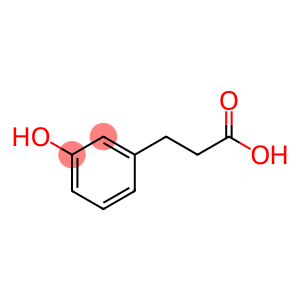
3-(3-Hydroxyphenyl)propionic acid
CAS: 621-54-5
Molecular Formula: C9H10O3
621-54-5 - Names and Identifiers
| Name | 3-(3-Hydroxyphenyl)propionic acid |
| Synonyms | IFLAB-BB F2119-0005 M-HYDROCOUMARIC ACID LABOTEST-BB LT00454600 3-Hydroxyhydrocinnamic acid 3-HYDROXYHYDROCINNAMIC ACID 3-(3-hydroxyl phenyl)propanol 3-hydroxy-benzenepropanoicaci 3-Hydroxyphenyl-propionic acid 3-(3-Hydroxyphenyl)propionic acid 3-(3-hydroxyphenyl)propanoic acid 3-(3-HYDROXYPHENYL)PROPIONIC ACID 3-(M-HYDROXYPHENYL)PROPIONIC ACID |
| CAS | 621-54-5 |
| EINECS | 210-692-8 |
| InChI | InChI=1/C9H10O3/c10-8-3-1-2-7(6-8)4-5-9(11)12/h1-3,6,10H,4-5H2,(H,11,12) |
621-54-5 - Physico-chemical Properties
| Molecular Formula | C9H10O3 |
| Molar Mass | 166.17 |
| Density | 1.260 |
| Melting Point | 111 °C |
| Boling Point | 355℃ |
| Flash Point | 182℃ |
| Solubility | Soluble in water, ethanol, ether |
| Vapor Presure | 1.23E-05mmHg at 25°C |
| Appearance | White-like to white crystals. |
| Color | Pale Beige to Light Brown |
| BRN | 1947445 |
| pKa | 4.68±0.10(Predicted) |
| Storage Condition | Sealed in dry,Room Temperature |
| Refractive Index | 1.58 |
| MDL | MFCD00016554 |
| Physical and Chemical Properties | Off-white to white crystals. |
621-54-5 - Risk and Safety
| Risk Codes | 36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. |
| RTECS | MW5340000 |
| HS Code | 29163990 |
621-54-5 - Reference
| Reference Show more | 1. [IF=4.952] Yuxin Hao et al."Stability and mechanism of phenolic compounds from raspberry extract under in vitro gastrointestinal digestion."Lwt Food Sci Technol. 2021 Mar;139:110552 2. [IF=4.24] Nana Li et al."Characterization of phenolic compounds and anti-acetylcholinase activity of coconut shells."Food Biosci. 2021 Aug;42:101204 3. [IF=4.952] Yulong Wei et al."Characterization of blueberry (Vaccinium corymbosum L.) catechol oxidases III binding mechanism in response to selected substrates and inhibitors."Lwt Food Sci Technol. 2022 Mar;158:113142 |
621-54-5 - Reference Information
| biological activity | 3-(3-Hydroxyphenyl)propionic acid (m-Hydroxyphenylpropionic acid, 3-hydroxyhydroxy acid) in the colon is a major metabolite of caffeic acid intake and is a phenolic metabolite of proanthocyanidins degraded by microorganisms. |
Last Update:2024-04-09 20:52:54
Supplier List
Product Name: 3-(3-HYDROXYPHENYL)PROPIONIC ACID Request for quotation
CAS: 621-54-5
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 621-54-5
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Multiple SpecificationsSpot supply
Product Name: 3-(3-Hydroxyphenyl)propanoic acid Visit Supplier Webpage Request for quotationCAS: 621-54-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Multiple SpecificationsSpot supply
Product Name: 3-(3-Hydroxyphenyl)Propionic Acid Visit Supplier Webpage Request for quotationCAS: 621-54-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: 3-(3-HYDROXYPHENYL)PROPIONIC ACID Request for quotation
CAS: 621-54-5
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 621-54-5
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Multiple SpecificationsSpot supply
Product Name: 3-(3-Hydroxyphenyl)propanoic acid Visit Supplier Webpage Request for quotationCAS: 621-54-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Multiple SpecificationsSpot supply
Product Name: 3-(3-Hydroxyphenyl)Propionic Acid Visit Supplier Webpage Request for quotationCAS: 621-54-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


