6119-47-7
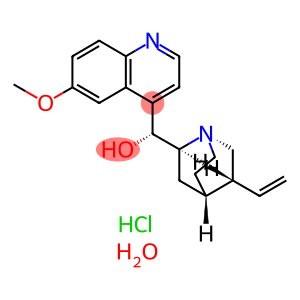
Quinine monohydrochloride dihydrate
CAS: 6119-47-7
Molecular Formula: C20H27ClN2O3
6119-47-7 - Names and Identifiers
| Name | Quinine monohydrochloride dihydrate |
| Synonyms | Quinine Monohy Quinine HCl Dihydrate Quinine monohydrochloride dihydrate QUININEHYDROCHLORIDE,DIHYDRATE,CRYSTAL (9R)-6'-methoxycinchonan-1'-ium-9-ol chloride dihydrate (8α, 9R)-6′-Methoxycinchonan-9-ol monohydrochloride dihydrate (4beta,8alpha,9R)-6'-methoxycinchonan-9-ol hydrochloride dihydrate Cinchonan-9-ol, 6-methoxy-, monohydrochloride, dihydrate, (8.alpha.,9R)- (R)(6-Methoxy-4-quinolyl)(5-vinyl-1-azabicyclo[2.2.2]oct-2-yl)methanol hydrochloride (R)(6-methoxy-4-quinolyl)(5-vinyl-1-azabicyclo[2.2.2]oct-2-yl)methanol monohydrochloride dihydrate |
| CAS | 6119-47-7 |
| EINECS | 612-097-2 |
| InChI | InChI=1/C20H24N2O2.ClH.2H2O/c1-3-13-12-22-9-7-14(13)10-19(22)20(23)16-6-8-21-18-5-4-15(24-2)11-17(16)18;;;/h3-6,8,11,13-14,19-20,23H,1,7,9-10,12H2,2H3;1H;2*1H2/t13?,14?,19?,20-;;;/m1.../s1 |
| InChIKey | MPQKYZPYCSTMEI-HPVDNOPASA-N |
6119-47-7 - Physico-chemical Properties
| Molecular Formula | C20H27ClN2O3 |
| Molar Mass | 378.9 |
| Melting Point | 115-116°C (dec.)(lit.) |
| Boling Point | 495.9°C at 760 mmHg |
| Specific Rotation(α) | [α]D20 -245~-255° (c=2, dil. HCl) (After Drying) |
| Flash Point | 122 °C |
| Water Solubility | Sparingly soluble in water.(0.26 g/L) (25°C), |
| Solubility | H2O: soluble0.25/5g/mL, clear, colorless |
| Vapor Presure | 1.19E-10mmHg at 25°C |
| Appearance | Crystalline Powder or Silky Needles |
| Color | White to off-white |
| Merck | 14,8061 |
| BRN | 6112655 |
| PH | 6.0 (10g/l, H2O, 20℃) |
| Storage Condition | Keep in dark place,Inert atmosphere,2-8°C |
| Sensitive | Light Sensitive |
| Refractive Index | -250 ° (C=2, EtOH) |
| MDL | MFCD00151248 |
| Use | K channel blocker. |
| In vitro study | Quinine blocked the Cx36 and Cx50 junction currents in a reversible and concentration-dependent manner, with half-maximal blocking concentrations of 32 mM and 73 mM, respectively. Quinine induces a slow transition between the open and fully closed states, which reduces the open probability of the channel. Quinine prevents certain types of gap junction channels, including those between neurons, from forming through CX36. Quinine, a K-channel blocker, prevents the formation of tumor necrosis factor (TNF) and subsequent liver DNA fragmentation and liver enzyme leakage. Quinine caused a Fos-like immune response (FLI) to concentrate in the medial third nucleus; Acid caused a more widespread FLI concentration more laterally. Quinine had a relatively weak effect on doxorubicin accumulation but completely restored doxorubicin sensitivity in resistant cells. Quinine also modified intracellular accumulation of doxorubicin, suggesting a modified intracellular drug distribution. Quinine predominantly blocks the voltage dependence in the whole-cell potassium current (IK). Quinine has also been used to dependently reduce sodium current (INa) while leaving calcium current (ICA) relatively unaffected. |
6119-47-7 - Risk and Safety
| Risk Codes | R22 - Harmful if swallowed R42/43 - May cause sensitization by inhalation and skin contact. R36/37/38 - Irritating to eyes, respiratory system and skin. R20/21/22 - Harmful by inhalation, in contact with skin and if swallowed. |
| Safety Description | S22 - Do not breathe dust. S36/37 - Wear suitable protective clothing and gloves. S45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36 - Wear suitable protective clothing. |
| UN IDs | 1544 |
| WGK Germany | 3 |
| RTECS | VA7700000 |
| FLUKA BRAND F CODES | 8 |
| HS Code | 29392000 |
| Hazard Note | Irritant |
| Toxicity | LD50 orally in Rabbit: 620 mg/kg |
6119-47-7 - Introduction
Solubility in water: 62.5g/l (20 C), soluble in ethanol, glycerol and chloroform, slightly soluble in ether.
Last Update:2022-10-16 17:27:06
Supplier List
Spot supply
Product Name: Quinine hydrochloride dihydrate Visit Supplier Webpage Request for quotationCAS: 6119-47-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: Quinine Visit Supplier Webpage Request for quotationCAS: 6119-47-7
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Product Name: Quinine monohydrochloride dihydrate Request for quotation
CAS: 6119-47-7
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 6119-47-7
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: Quinine hydrochloride dihydrate Request for quotation
CAS: 6119-47-7
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 6119-47-7
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Spot supply
Product Name: Quinine hydrochloride dihydrate Visit Supplier Webpage Request for quotationCAS: 6119-47-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Quinine hydrochloride dihydrate Visit Supplier Webpage Request for quotationCAS: 6119-47-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: Quinine Visit Supplier Webpage Request for quotationCAS: 6119-47-7
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Product Name: Quinine monohydrochloride dihydrate Request for quotation
CAS: 6119-47-7
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 6119-47-7
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: Quinine hydrochloride dihydrate Request for quotation
CAS: 6119-47-7
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 6119-47-7
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Spot supply
Product Name: Quinine hydrochloride dihydrate Visit Supplier Webpage Request for quotationCAS: 6119-47-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History




