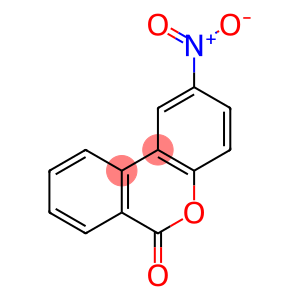
6-NITRO-3,4-BENZOCOUMARIN
6-nitro-3,4-benzocoumarin
CAS: 6623-66-1
Molecular Formula: C13H7NO4
6-NITRO-3,4-BENZOCOUMARIN - Names and Identifiers
| Name | 6-nitro-3,4-benzocoumarin |
| Synonyms | 2-NITRODIBENZOPYRANONE 6-NITRO-3,4-BENZOCOUMARIN 6-nitro-3,4-benzocoumarin d)pyran-6-one,2-nitro-6h-dibenzo( 2-nitro-6h-dibenzo(b,d)pyran-6-one 6H-Dibenzo[b,d]pyran-6-one, 2-nitro- 2'-hydroxy-5'-nitro-2-biphenylcarboxylicacidelta-lactone |
| CAS | 6623-66-1 |
| InChI | InChI=1/C22H20BrN3S/c23-21-10-9-16(27-21)15-24-26-13-11-25(12-14-26)22-19-7-3-1-5-17(19)18-6-2-4-8-20(18)22/h1-10,15,22H,11-14H2 |
6-NITRO-3,4-BENZOCOUMARIN - Physico-chemical Properties
| Molecular Formula | C13H7NO4 |
| Molar Mass | 241.2 |
| Density | 1.3304 (rough estimate) |
| Melting Point | 259-261°C (dec.)(lit.) |
| Boling Point | 383.97°C (rough estimate) |
| Flash Point | 303.2°C |
| Vapor Presure | 2.4E-13mmHg at 25°C |
| Refractive Index | 1.4800 (estimate) |
6-NITRO-3,4-BENZOCOUMARIN - Risk and Safety
| Hazard Symbols | Xi - Irritant |
| Risk Codes | 36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S37/39 - Wear suitable gloves and eye/face protection |
| WGK Germany | 3 |
| RTECS | HP8757550 |
6-NITRO-3,4-BENZOCOUMARIN - Introduction
6-Nitro-3, 4-Benzocoumarin (6-Nitro-3, 4-Benzocoumarin) is an organic compound. It has the following properties:
1. Appearance: 6-nitro-3,4-benzocoumarin is a pale yellow to yellow crystalline solid.
2. melting point: its melting point range of 191-194 ℃.
Here are its uses:
1. Fluorescent dye: 6-nitro-3,4-benzocoumarin can be used as fluorescent probe and dye for biological imaging research and drug analysis.
2. Photosensitive compound: It can also be used as a photosensitizer for photochemical reactions and photocatalytic reactions.
3. Pharmaceutical field: Some studies have shown that 6-nitro-3, 4-benzocumarin has anti-tumor activity and may have potential applications in drug development.
The method of preparing 6-nitro-3,4-benzocoumarin includes the following steps:
1. First, a nitro group is introduced through a substitution reaction on the benzene ring.
2. Then, the compound is reacted with coumarone for a ring-closing reaction to form the target product.
Safety Information:
There is limited safety data for 6-nitro-3,4-benzocoumarin. Because its effects on humans and the environment have not been fully assessed, standard laboratory safety practices should be followed when using and handling this compound. Wear personal protective equipment, such as goggles and gloves, if necessary. Also, the compound should be stored and disposed of properly to avoid reaction with other chemicals.
1. Appearance: 6-nitro-3,4-benzocoumarin is a pale yellow to yellow crystalline solid.
2. melting point: its melting point range of 191-194 ℃.
Here are its uses:
1. Fluorescent dye: 6-nitro-3,4-benzocoumarin can be used as fluorescent probe and dye for biological imaging research and drug analysis.
2. Photosensitive compound: It can also be used as a photosensitizer for photochemical reactions and photocatalytic reactions.
3. Pharmaceutical field: Some studies have shown that 6-nitro-3, 4-benzocumarin has anti-tumor activity and may have potential applications in drug development.
The method of preparing 6-nitro-3,4-benzocoumarin includes the following steps:
1. First, a nitro group is introduced through a substitution reaction on the benzene ring.
2. Then, the compound is reacted with coumarone for a ring-closing reaction to form the target product.
Safety Information:
There is limited safety data for 6-nitro-3,4-benzocoumarin. Because its effects on humans and the environment have not been fully assessed, standard laboratory safety practices should be followed when using and handling this compound. Wear personal protective equipment, such as goggles and gloves, if necessary. Also, the compound should be stored and disposed of properly to avoid reaction with other chemicals.
Last Update:2024-04-09 21:11:58