57-71-6
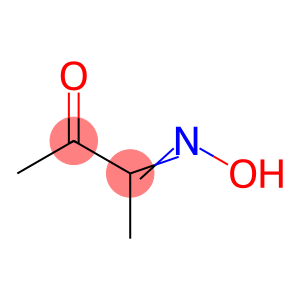
2,3-Butanedione monoxime
CAS: 57-71-6
Molecular Formula: C4H7NO2
57-71-6 - Names and Identifiers
57-71-6 - Physico-chemical Properties
| Molecular Formula | C4H7NO2 |
| Molar Mass | 101.1 |
| Density | 1.2085 (rough estimate) |
| Melting Point | 75-78°C(lit.) |
| Boling Point | 185-186°C(lit.) |
| Flash Point | 185-186°C |
| Water Solubility | 5 g/100 mL (20 ºC) |
| Solubility | Chloroform (Slightly), Methanol (Slightly) |
| Vapor Presure | 0.315mmHg at 25°C |
| Appearance | White or white-like crystals |
| Color | White to almost white |
| Odor | Odorless |
| BRN | 605582 |
| pKa | 9.32±0.10(Predicted) |
| Storage Condition | Inert atmosphere,Room Temperature |
| Stability | Stable. Incompatible with strong oxidizing agents. |
| Sensitive | Hygroscopic |
| Refractive Index | 1.4340 (estimate) |
| MDL | MFCD00002116 |
| Physical and Chemical Properties | Melting Point: 75-78°C Boiling Point: 185-186°C |
| Use | Used as a developer for qualitative inspection of Ni2 and photometric determination of Ni2, Co, Pt4 and Pd2, etc., and also used for photometric determination of urea and acylurea. Pharmaceutical intermediates. |
57-71-6 - Risk and Safety
| Hazard Symbols | Xn - Harmful |
| Risk Codes | R36/37/38 - Irritating to eyes, respiratory system and skin. R20/21/22 - Harmful by inhalation, in contact with skin and if swallowed. |
| Safety Description | S22 - Do not breathe dust. S24/25 - Avoid contact with skin and eyes. S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. |
| WGK Germany | 3 |
| RTECS | EK3150000 |
| TSCA | Yes |
| HS Code | 29280090 |
| Toxicity | LD50 ipr-mus: 51 mg/kg JPMSAE 53,1143,64 |
57-71-6 - Introduction
Slightly soluble in water. Soluble in ethanol, ether, acetone and chloroform. It forms light yellow or orange complexes with Ni2, Pd2, Co2 and ReO, which are easy to be extracted by chloroform.
Last Update:2022-10-16 17:26:30
57-71-6 - Reference Information
| NIST chemical information | information provided by: webbook.nist.gov (external link) |
| EPA chemical substance information | information provided by: ofmpeb.epa.gov (external link) |
| Background | cold cardioplegia is commonly used in cardiac surgery to protect the myocardium and reduce ischemic injury. At present, two types of crystalloid cardioplegia are commonly used, namely, intracellular and extracellular cardioplegia, both of which can reduce myocardial energy consumption and delay irreversible myocardial injury. Recently, 2, 3-butanedione monooxime (BDM), a drug that uncouples cardiac myosin to actin as the "third concept" of myocardial protection, more and more attention. Some studies have confirmed that BDM can increase the tolerance of rabbit, rat and human hearts to cold ischemia, promote the inactivation of L-type Ca2 channels and reduce myocardial energy consumption. |
| Application | 2, 3-butanedione oxime is used for the preparation of the sodium and potassium salts of butanedione monooxime. Butanedione monooxime sodium, potassium salt is a highly toxic chemical Disinfection agent of the main active ingredients, as the main agent of the skin Disinfection agent with the traditional skin Disinfection agent does not have the non-toxic, no stimulation, easy storage, corrosion, Disinfection of the advantages of fast speed. |
| Use | pharmaceutical intermediates. Reagents were analyzed. It is used as a developer for qualitative examination of Ni2 and Photometric Determination of Ni2, Co, Pt4 and Pd2, etc., and also used for Photometric Determination of urea and ureide. Pharmaceutical intermediates. |
| production method | is obtained by reacting methyl ethyl ketone with ethyl nitrite. Methyl Ethyl Ketone and hydrochloric acid were added to the reaction pan, and ethyl nitrite gas was immediately passed through. The reaction was maintained at 40-55 °c until the gas passed. The ethanol produced by the reaction was distilled off under reduced pressure, and the distillation was stopped at 90 ° C. To obtain the butanedione monoxime. The yield was 82-87%. |
Last Update:2024-04-10 22:29:15
Supplier List
Multiple SpecificationsSpot supply
Product Name: Diacetylmonoxime Visit Supplier Webpage Request for quotationCAS: 57-71-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2,3-Butanedione monoxime Request for quotation
CAS: 57-71-6
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 57-71-6
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: 2,3-Butanedione monoxime Request for quotation
CAS: 57-71-6
Tel: 13247110337
Email: 2205437118@qq.com
Mobile: 13247110337
QQ: 2205437118
Wechat: 13247110337
CAS: 57-71-6
Tel: 13247110337
Email: 2205437118@qq.com
Mobile: 13247110337
QQ: 2205437118
Wechat: 13247110337
Multiple Specifications
Product Name: Diacetyl monoxime Visit Supplier Webpage Request for quotationCAS: 57-71-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Multiple SpecificationsSpot supply
Product Name: Diacetylmonoxime Visit Supplier Webpage Request for quotationCAS: 57-71-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2,3-Butanedione monoxime Request for quotation
CAS: 57-71-6
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 57-71-6
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: 2,3-Butanedione monoxime Request for quotation
CAS: 57-71-6
Tel: 13247110337
Email: 2205437118@qq.com
Mobile: 13247110337
QQ: 2205437118
Wechat: 13247110337
CAS: 57-71-6
Tel: 13247110337
Email: 2205437118@qq.com
Mobile: 13247110337
QQ: 2205437118
Wechat: 13247110337
Multiple Specifications
Product Name: Diacetyl monoxime Visit Supplier Webpage Request for quotationCAS: 57-71-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



