569-57-3
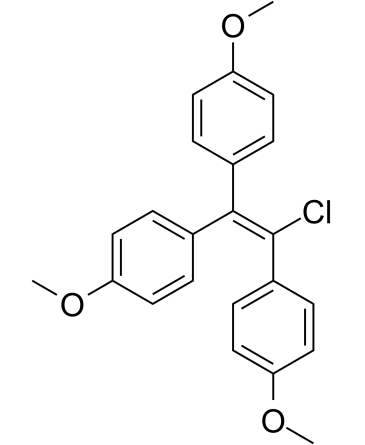
CHLOROTRIANISENE
CAS: 569-57-3
Molecular Formula: C23H21ClO3
569-57-3 - Names and Identifiers
| Name | CHLOROTRIANISENE |
| Synonyms | CHLOROTRIANISENE Chlortrianisoestrol Chlorotrianisene (1 g) chlorotris(4-methoxyphenyl)ethylene 1-Chloro-1,2,2-tris(4-methoxyphenyl)ethene 1,1',1''-(1-Chloro-1,2,2-ethenetriyl)tris[4-methoxybenzene] 1-[2-chloro-1,2-bis(4-methoxyphenyl)ethenyl]-4-methoxybenzene 1-[2-chloro-1,2-bis(4-methoxyphenyl)ethenyl]-4-methoxy-benzene |
| CAS | 569-57-3 |
| EINECS | 209-318-6 |
569-57-3 - Physico-chemical Properties
| Molecular Formula | C23H21ClO3 |
| Molar Mass | 380.86 |
| Density | 1.1010 (rough estimate) |
| Melting Point | 114-116° |
| Boling Point | 496.07°C (rough estimate) |
| Appearance | Solid powder |
| Storage Condition | -20℃ |
| Refractive Index | 1.4585 (estimate) |
| Physical and Chemical Properties | Chemical properties Melting point 114-116 ℃, solubility in ethanol 0.28g/100ml, solubility in ether 3.6g/100ml, soluble in acetone, chloroform, carbon tetrachloride and benzene, insoluble in water. |
| Use | Use of estrogen drugs, mainly for the treatment of prostate enlargement and women menopausal syndrome. |
569-57-3 - Introduction
569-57-3 - Uses and synthesis methods
pharmacological action
Chlorene estradiol is a non-steroidal estrogen drug. Its activity is weaker than diethylstilbestrol. It can regulate the release of gonadotropins in the anterior pituitary. By reducing the release of hypothalamic luteinizing hormone release factor, It leads to the reduction of follicle-stimulating hormone (FSH) and the release of luteinizing hormone (LH) from the pituitary gland. Because it causes hyperfunction, long-term use will not cause pituitary enlargement and adrenal hyperplasia, the effect is relatively mild, and the human body can tolerate it well.
Pharmacokinetics
after taking this product, it can be stored in adipose tissue and released slowly. it is metabolized by liver to substances containing estrogen, so it is called estrogen precursor. This product is metabolized in the liver after administration, mainly excreted in feces, and its active substances can be detected. Rats take 5mg orally, which can also be detected in feces on the 10th day. After 70mg orally, monkeys still have 6% ~ 18% estrogen active substances on the 30th day.
indications
This product is used to treat women's menopausal syndrome and symptoms caused by estrogen deficiency after surgery; adolescent dysfunctional uterine bleeding; estrogen replacement therapy for women's gonadal insufficiency; male prostate hyperplasia.
biological activity
Chlorotrianisene (tri-p-anisylchloroethylene, TACE, CTA) is a synthetic, non-steroidal estrogen. It is a selective estrogen receptor modulator.
target
Target Value
Estrogen receptor
in vitro study
Comparison of intracellular estrogen receptor (ER) affinities of Chlorotrianisene with respective rat uterine cytosolic ER affinities has initially suggested the potential for activation of ER as a mechanism of growth stimulation. Chlorotrianisene exhibits concentration dependent cell growth stimulation with an EC 50 of 28 nM and a K I of 500 nM in MCF-7 cells.
in vivo studies
The incubation of Chlorotrianisene with rat liver microsomes and NADPH generates a reactive intermediate which binds covalently to proteins. Intermediate may inactivate the uterine estrogen receptors (ER). The incubation of Chlorotrianisene with rat liver microsomes and NADPH in the presence of rat uteri, under conditions which generate intermediate, markedly decreased the binding capacity of the ER for [ 3 H]estradiol (E2).
production method
There are many synthetic routes, which are obtained by bromination, salt formation, condensation, and chlorination of p-methoxyacetophenone. The total yield is 14.5%; it can also be prepared from anisic acid, the total yield is 44.4%, but the price of anisic acid raw material is more expensive. Using anisole as raw material, Taishu was prepared by the following steps with a total yield of 21.6%. 1. Preparation of chlorinated p-methoxyacetophenone Anisole reacts with chloroacetyl chloride to obtain chlorinated p-methoxyacetophenone. 2.1,1,2-tris (p-methoxyphenyl) ethylene Preparation of bromoanisole and magnesium to obtain Grignard reagent, and reacts with chlorinated p-methoxyacetophenone to obtain oil and continues to react with PTS to obtain 1,1,2-tris (p-methoxyphenyl) ethylene. 3. Preparation of Tetris 1,1,2-tris (p-methoxyphenyl) ethylene chloride Tetris.
Supplier List
CAS: 569-57-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 569-57-3
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 569-57-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 569-57-3
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565

