5650-44-2
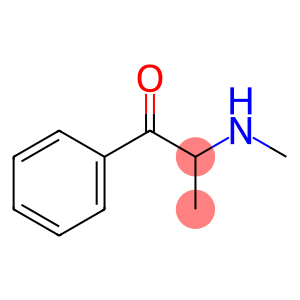
2-(methylamino)propiophenone
CAS: 5650-44-2
Molecular Formula: C10H13NO
5650-44-2 - Names and Identifiers
5650-44-2 - Physico-chemical Properties
| Molecular Formula | C10H13NO |
| Molar Mass | 163.22 |
| Density | 1.000±0.06 g/cm3(Predicted) |
| Melting Point | 182-184 °C |
| Boling Point | 254.8±23.0 °C(Predicted) |
| pKa | 7.14±0.10(Predicted) |
5650-44-2 - References
| EPA chemical substance information | information provided by: ofmpeb.epa.gov (external link) |
| pharmacological action | methcathinone, as a semi-synthetic compound, belongs to the derivatives of cathinones. The cathinone is a kind of natural Amphetamine alkaloids from the stem and leaf of plant khat. Methcathinone, like other derivatives of cathinones, belongs to the group β-keto-amphetamine. As an amphetamine-type central nervous system stimulant, its pharmacological effects may be achieved by promoting the release of monoamine transmitters and inhibiting reuptake. In an in vitro study, methcathinone and another cathinone derivative, 2-methylamino-1-propanone, were found to have a very similar mechanism to methamphetamine, the main characteristic is that it acts on the transport of catecholamines on the plasma membrane, and the difference is that the relative binding force of norepinephrine, dopamine and serotonin is different when transporting. Common forms of abuse of methcathinone are nasal and oral, and abuse has also been reported with intravenous or rectal administration via capsules. According to abusers, the inhalation of methcathinone can produce an effect within a few minutes, reaching a peak in about 30 minutes and lasting for 2-3 hours, while the duration of intravenous injection of drug effect will be shortened by nearly half. After absorption into the blood, due to the difference in chemical structure, compared with other β-keto-amphetamine substances, methcathinone has lower ability to cross the blood-brain barrier, but as a dopamine reuptake inhibitor, its pharmacological effects are similar to those of methamphetamine, and it is easy to cause euphoria and excitement, including: euphoria, increased alertness, dilated pupils, shortness of breath, increased heart rate, continuous talk, increased desire to communicate, increased sexual desire and cognitive impairment. The main metabolic mode of methcathinone is the oxidation of the tolyl group to the corresponding alcohol and carboxylic acid, so the appropriate urine detection technology can be used to detect methcathinone and other β-keto-amphetamine substances in the body. Figure 1 shows the chemical structure of methcathinone and related synthetic drugs |
| toxicological effects | manifestations of acute poisoning in abusers include irritability, Aggression behavior, tachycardia, confusion, chest pain, Nausea, palpitation, peripheral vascular contraction, Head Pain and other symptoms, there were 4 cases of convulsions, 1 cases of cardiac arrest, in addition, there were reports of side effects and serious adverse reactions of the sympathetic nervous system. A survey by the national addiction center in the United Kingdom found that 51 percent of methcathinone abusers had Head Pain, 43 percent had palpitations, and 27 percent felt Nausea, 15% of the patients presented with cold or blue-purple toxic symptoms and signs indicating peripheral capillary contraction. There are also studies in the UK that have found that methcathinone can induce necrotizing vasculitis, segmental glomerulonephritis, and Henoch-Schonlein purpura and nephritis in children. Since the prevalence of methcathinone abuse is not long, the results of its chronic toxicity studies are still unclear. In animal studies, researchers have demonstrated that methcathinone is toxic to dopaminergic neurons in rats and point out that this toxicity is related to its chemical nature and is not caused by the manganese impurity contained in the compound. A case report of an abuser who used methcathinone twice a week for 18 months developed dependence due to hallucinations, agitation, he was admitted to the hospital for treatment of psychotic symptoms such as excitement and mania. Given the similarity of methcathinone and cathinone, some researchers believe that long-term use may also lead to sexual dysfunction. The special toxicity of methcathinone (teratogenic, carcinogenic, mutagenic) has not been reported. As of July 2010, a total of 52 deaths related to the abuse of methcathinone have been reported in the UK, of which 2 have been confirmed to be due to direct abuse of methcathinone. The cause of death in the remaining cases is mostly due to the underlying disease of the abuser or the combination of other substances, but studies have shown that methcathinone can increase the abuse of tobacco, craving and dependence on alcohol and other illicit drugs. |
Last Update:2024-04-09 02:00:23
Supplier List
Product Name: *Cathinone Impurity 2 (Methcathinone) Request for quotation
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
View History
