541547-37-9
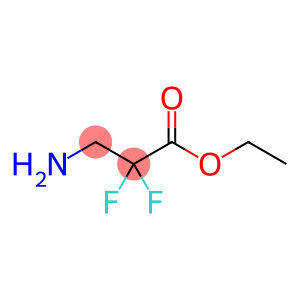
Ethyl 2,2-difluoro-3-amino-propanoate hydrochloride
CAS: 541547-37-9
Molecular Formula: C5H9F2NO2
541547-37-9 - Names and Identifiers
541547-37-9 - Physico-chemical Properties
| Molecular Formula | C5H9F2NO2 |
| Molar Mass | 153.13 |
| Density | 1.185 |
| Boling Point | 195℃ |
| Flash Point | 72℃ |
| pKa | 6.19±0.30(Predicted) |
| Storage Condition | Room Temprature |
| MDL | MFCD13185039 |
541547-37-9 - Introduction
Ethyl 2, hydrochloride is an organic compound with the formula C5H9ClF2NO2. The following are some properties, uses, preparation and safety information about the compound:
Nature:
1. Appearance: It is a white crystalline solid.
2. melting point: its melting point range of about 85-90 ℃.
3. Solubility: It has low solubility in water, but it is soluble in some organic solvents, such as alcohols and esters.
4. chemical properties: it is a weak alkaline substance.
Use:
Ethyl 2, hydrochloride has certain applications in chemical laboratories and the pharmaceutical industry:
1. As an intermediate, it can be used to synthesize other organic compounds.
2. It can be used in organic synthesis reactions, such as olefins and haloalkylation reagents.
3. can be used as chemical raw materials for drug research and development.
Preparation Method:
Ethyl 2, the hydrochloride can be synthesized by the following steps:
1. First, ethyl 2,2-difluoro-3-aminopropionate is reacted with hydrochloric acid to generate Ethyl 2, an intermediate of hydrochloride.
2. Then, through crystallization or other purification methods, pure Ethyl 2, hydrochloride, can be obtained from the reaction product.
Safety Information:
1. Ethyl 2, hydrochloride may be irritating to eyes, skin and respiratory tract. Use should wear appropriate protective equipment to avoid contact.
2. When using or handling the compound, the relevant safety procedures should be followed and good ventilation conditions should be ensured.
3. During storage, it should be kept in a sealed container, away from heat and fire sources, and avoid contact with oxidants.
4. When disposing of waste from this compound, follow local regulations and dispose of it appropriately.
Nature:
1. Appearance: It is a white crystalline solid.
2. melting point: its melting point range of about 85-90 ℃.
3. Solubility: It has low solubility in water, but it is soluble in some organic solvents, such as alcohols and esters.
4. chemical properties: it is a weak alkaline substance.
Use:
Ethyl 2, hydrochloride has certain applications in chemical laboratories and the pharmaceutical industry:
1. As an intermediate, it can be used to synthesize other organic compounds.
2. It can be used in organic synthesis reactions, such as olefins and haloalkylation reagents.
3. can be used as chemical raw materials for drug research and development.
Preparation Method:
Ethyl 2, the hydrochloride can be synthesized by the following steps:
1. First, ethyl 2,2-difluoro-3-aminopropionate is reacted with hydrochloric acid to generate Ethyl 2, an intermediate of hydrochloride.
2. Then, through crystallization or other purification methods, pure Ethyl 2, hydrochloride, can be obtained from the reaction product.
Safety Information:
1. Ethyl 2, hydrochloride may be irritating to eyes, skin and respiratory tract. Use should wear appropriate protective equipment to avoid contact.
2. When using or handling the compound, the relevant safety procedures should be followed and good ventilation conditions should be ensured.
3. During storage, it should be kept in a sealed container, away from heat and fire sources, and avoid contact with oxidants.
4. When disposing of waste from this compound, follow local regulations and dispose of it appropriately.
Last Update:2024-04-09 21:01:54
Supplier List
Product Name: Ethyl 2,2-difluoro-3-amino-propanoate hydrochloride Request for quotation
CAS: 541547-37-9
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 541547-37-9
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: Ethyl 2,2-difluoro-3-amino-propanoate hydrochloride Request for quotation
CAS: 541547-37-9
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 541547-37-9
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
View History
