52364-73-5
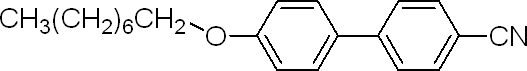
4'-(Octyloxy)-4-biphenylcarbonitrile
CAS: 52364-73-5
Molecular Formula: C21H25NO
52364-73-5 - Names and Identifiers
| Name | 4'-(Octyloxy)-4-biphenylcarbonitrile |
| Synonyms | 4'-(Octyloxy)-4-cyanobiphenyl 4'-(n-Octyloxy)-4-cyanobiphenyl 4-(4-octyloxyphenyl)benzonitrile 4'-(Octyloxy)-4-biphenylcarbonitrile 1'-biphenyl]-4-carbonitrile,4'-(octyloxy)-[ 4'-(Octyloxy)[1,1'-biphenyl]-4-carbonitrile 1'-biphenyl)-4-carbonitrile,4'-(octyloxy)-( 1'-Biphenyl]-4-carbonitrile,4'-(octyloxy)-[1 [1,1'-Biphenyl]-4-carbonitrile, 4'-(octyloxy)- |
| CAS | 52364-73-5 |
| EINECS | 257-878-5 |
52364-73-5 - Physico-chemical Properties
| Molecular Formula | C21H25NO |
| Molar Mass | 307.43 |
| Density | 1.0505 (rough estimate) |
| Melting Point | 51-77 °C (lit.) |
| Boling Point | 447.93°C (rough estimate) |
| Flash Point | >230°F |
| Appearance | Morphological liquid crystal (Nematic) |
| Storage Condition | Sealed in dry,Room Temperature |
| Refractive Index | 1.5614 (estimate) |
| MDL | MFCD00075145 |
| Use | Application of p-cyanoxybiphenyl can be used as organic synthesis intermediates and pharmaceutical intermediates, mainly used in laboratory research and development process and chemical production process. |
52364-73-5 - Risk and Safety
| Risk Codes | R20/21/22 - Harmful by inhalation, in contact with skin and if swallowed. R36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36 - Wear suitable protective clothing. |
| WGK Germany | 3 |
| RTECS | DV1764500 |
| TSCA | Yes |
| HS Code | 29269090 |
52364-73-5 - Upstream Downstream Industry
52364-73-5 - Nature
| NIST chemical information | Octyloxycyanobiphenyl(52364-73-5) |
| EPA chemical information | [1,1'-Biphenyl]-4-carbonitrile, 4'-(octyloxy)- (52364-73-5) |
Last Update:2024-04-10 22:29:15
52364-73-5 - Reference Information
| application | p-cyanooctyloxybiphenyl can be used as organic synthesis intermediates and pharmaceutical intermediates, mainly used in laboratory research and development processes and chemical production processes. |
| preparation | 1-bromooctane (20 mmol, 3.5 mL) was added dropwise under nitrogen to the atmosphere of a stirred mixture of 4 '-hydroxy -4-dibenzonitrile (15.4 mmol, 3g) and anhydrous K2CO3 (14.5 mmol, 2g) in 200 mL anhydrous DMSO. Heat the reaction mixture at 110°C for 3 hours. The reaction mixture was added dropwise to 400 mL of 10% NaOH solution at room temperature and filtered. The resulting mixture was vacuum dried and crystallized from ethanol. The product was vacuum dried to obtain p-cyanooctyloxybiphenyl. |
| NIST chemical information | Information provided by: webbook.nist.gov (external link) |
| EPA chemical information | Information provided by: ofmpub.epa.gov (external link) |
Last Update:2024-04-10 22:29:15
52364-73-5 - Preparation
1-bromooctane (20 mmol, 3.5 mL) was added dropwise under nitrogen to a stirred mixture of 4 '-hydroxy-4-bibenzonitrile (15.4 mmol, 3g) and anhydrous K2CO3 (14.5 mmol, 2g) in 200 mL anhydrous DMSO in the atmosphere. The reaction mixture was heated at 110°C for 3 hours. The reaction mixture was added dropwise to 400 mL of 10% NaOH solution at room temperature and filtered. The resulting mixture was vacuum dried and crystallized from ethanol. The product was vacuum dried to obtain p-cyanooctyloxybiphenyl.
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: Octyloxycyanobiphenyl Visit Supplier Webpage Request for quotationCAS: 52364-73-5
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: 4′-(Octyloxy)-4-biphenylcarbonitrile Visit Supplier Webpage Request for quotationCAS: 52364-73-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 4'-(Octyloxy)-4-biphenylcarbonitrile Request for quotation
CAS: 52364-73-5
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 52364-73-5
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Spot supply
Product Name: 4′-(Octyloxy)-4-biphenylcarbonitrile Visit Supplier Webpage Request for quotationCAS: 52364-73-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Octyloxycyanobiphenyl Visit Supplier Webpage Request for quotationCAS: 52364-73-5
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: 4′-(Octyloxy)-4-biphenylcarbonitrile Visit Supplier Webpage Request for quotationCAS: 52364-73-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 4'-(Octyloxy)-4-biphenylcarbonitrile Request for quotation
CAS: 52364-73-5
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 52364-73-5
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Spot supply
Product Name: 4′-(Octyloxy)-4-biphenylcarbonitrile Visit Supplier Webpage Request for quotationCAS: 52364-73-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History
Raw Materials for 52364-73-5



