50528-80-8
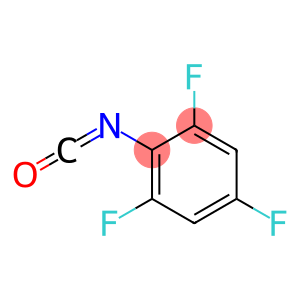
2,4,6-Trifluorophenyl isocyanate
CAS: 50528-80-8
Molecular Formula: C7H2F3NO
50528-80-8 - Names and Identifiers
| Name | 2,4,6-Trifluorophenyl isocyanate |
| Synonyms | 2,4,6-Trifluorophenyl isocyanate 2,4,6-TRIFLUOROPHENYL ISOCYANATE 1,3,5-trifluoro-2-isocyanatobenzene 1,3,5-Trifluoro-2-isocyanatobenzene Benzene, 1,3,5-trifluoro-2-isocyanato- |
| CAS | 50528-80-8 |
| EINECS | 680-441-9 |
| InChI | InChI=1/C7H2F3NO/c8-4-1-5(9)7(11-3-12)6(10)2-4/h1-2H |
50528-80-8 - Physico-chemical Properties
| Molecular Formula | C7H2F3NO |
| Molar Mass | 173.09 |
| Density | 1.35g/cm3 |
| Boling Point | 166.4°C at 760 mmHg |
| Flash Point | 52.2°C |
| Water Solubility | Hydrolyzes in water. |
| Vapor Presure | 1.79mmHg at 25°C |
| BRN | 6325663 |
| Storage Condition | Room Temprature |
| Sensitive | Moisture Sensitive |
| Refractive Index | 1.4760 |
| MDL | MFCD03426028 |
50528-80-8 - Risk and Safety
| Hazard Symbols | T - Toxic |
| Risk Codes | R23 - Toxic by inhalation R25 - Toxic if swallowed R36/37/38 - Irritating to eyes, respiratory system and skin. R42 - May cause sensitization by inhalation |
| Safety Description | S23 - Do not breathe vapour. S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. S45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) |
| UN IDs | 2206 |
| Hazard Note | Toxic |
| Hazard Class | 6.1 |
| Packing Group | III |
50528-80-8 - Introduction
2,4, 6-Trifluophenyl isocyanate is an organic compound with the chemical formula C7H2F3NO. The following is an introduction to its nature, use, preparation and safety information:
Nature:
2,4,6-Trifluorophenyl isocyanate is a colorless liquid with a pungent odor. It has a density of about 1.44 g/mL, a boiling point of about 112-113°C, and a melting point of about 18-19°C. It has good solubility in solvents, but is insoluble in water.
Use:
2,4,6-Trifluorophenyl isocyanate is often used in organic synthesis reactions as an important intermediate. It can be used in the preparation of other organic compounds such as pharmaceuticals, dyes and polymers. Its strong fluorine-based properties make it an important reagent in some chemical reactions.
Preparation Method:
2,4,6-Trifluorophenyl isocyanate is generally prepared by reacting 2,4, 6-trifluoroaniline with excess chloride and diethyl carbonate in the presence of a base. In this reaction, the chlorinating agent is used to chlorinate 2,4, 6-trifluoroaniline to form an intermediate having a chlorine atom, which is then reacted with diethyl carbonate to give 2,4, 6-Trifluorophene isocyanate via a base-catalyzed reaction.
Safety Information:
2,4,6-Trifluorophenyl isocyanate needs to be handled properly because it is an organic compound and has a certain degree of danger. During operation, appropriate personal protective equipment such as gloves, goggles and protective clothing should be worn. Avoid direct contact with skin and eyes, and operate in a well-ventilated area. Keep away from fire and high temperature to avoid fire and explosion hazards. Care should be taken to avoid contact with oxidants, strong acids and strong bases during storage and use.
Nature:
2,4,6-Trifluorophenyl isocyanate is a colorless liquid with a pungent odor. It has a density of about 1.44 g/mL, a boiling point of about 112-113°C, and a melting point of about 18-19°C. It has good solubility in solvents, but is insoluble in water.
Use:
2,4,6-Trifluorophenyl isocyanate is often used in organic synthesis reactions as an important intermediate. It can be used in the preparation of other organic compounds such as pharmaceuticals, dyes and polymers. Its strong fluorine-based properties make it an important reagent in some chemical reactions.
Preparation Method:
2,4,6-Trifluorophenyl isocyanate is generally prepared by reacting 2,4, 6-trifluoroaniline with excess chloride and diethyl carbonate in the presence of a base. In this reaction, the chlorinating agent is used to chlorinate 2,4, 6-trifluoroaniline to form an intermediate having a chlorine atom, which is then reacted with diethyl carbonate to give 2,4, 6-Trifluorophene isocyanate via a base-catalyzed reaction.
Safety Information:
2,4,6-Trifluorophenyl isocyanate needs to be handled properly because it is an organic compound and has a certain degree of danger. During operation, appropriate personal protective equipment such as gloves, goggles and protective clothing should be worn. Avoid direct contact with skin and eyes, and operate in a well-ventilated area. Keep away from fire and high temperature to avoid fire and explosion hazards. Care should be taken to avoid contact with oxidants, strong acids and strong bases during storage and use.
Last Update:2024-04-09 02:00:46
Supplier List
Spot supply
Product Name: 2,4,6-Trifluorophenyl isocyanate Visit Supplier Webpage Request for quotationCAS: 50528-80-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2,4,6-TRIFLUOROPHENYL ISOCYANATE Request for quotation
CAS: 50528-80-8
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 50528-80-8
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: 2,4,6-Trifluorophenyl isocyanate Visit Supplier Webpage Request for quotation
CAS: 50528-80-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 50528-80-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 2,4,6-Trifluorophenyl isocyanate Visit Supplier Webpage Request for quotationCAS: 50528-80-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2,4,6-TRIFLUOROPHENYL ISOCYANATE Request for quotation
CAS: 50528-80-8
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 50528-80-8
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: 2,4,6-Trifluorophenyl isocyanate Visit Supplier Webpage Request for quotation
CAS: 50528-80-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 50528-80-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


