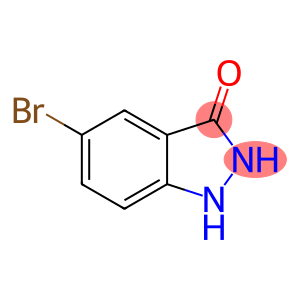
5-broMo-1,2-dihydro-3H-Indazol-3-one
5-bromo-1H-indazol-3-ol
CAS: 7364-27-4
Molecular Formula: C7H5BrN2O
5-broMo-1,2-dihydro-3H-Indazol-3-one - Names and Identifiers
| Name | 5-bromo-1H-indazol-3-ol |
| Synonyms | 5-Bromo-1H-indazol-3-ol 5-bromo-1H-indazol-3-ol 5-Bromoindazol-3(2H)-one 1H-indazol-3-ol, 5-bromo- 5-BROMO-3-HYDROXYINDAZOLE 5-BROMO-3-HYDROXY (1H)INDAZOLE 5-BROMO-1,2-DIHYDRO-INDAZOL-3-ONE 5-broMo-1,2-dihydro-3H-Indazol-3-one 3H-Indazol-3-one, 5-broMo-1,2-dihydro- 3H-Indazol-3-one, 5-bromo-1,2-dihydro- |
| CAS | 7364-27-4 |
| InChI | InChI=1/C7H5BrN2O/c8-4-1-2-6-5(3-4)7(11)10-9-6/h1-3H,(H2,9,10,11) |
5-broMo-1,2-dihydro-3H-Indazol-3-one - Physico-chemical Properties
| Molecular Formula | C7H5BrN2O |
| Molar Mass | 213.03 |
| Density | 1.728±0.06 g/cm3(Predicted) |
| Boling Point | 268.4±19.0 °C(Predicted) |
| Flash Point | 209.098°C |
| Vapor Presure | 0mmHg at 25°C |
| pKa | 11.02±0.20(Predicted) |
| Storage Condition | 2-8°C |
| Refractive Index | 1.784 |
5-broMo-1,2-dihydro-3H-Indazol-3-one - Introduction
5-bromo-1H-indazol-3-ol is an organic compound with the molecular formula C7H5BrN2O. The following is a description of its nature, use, preparation and safety information:
Nature:
-Appearance: 5-bromo-1H-indazol-3-ol is a white to pale yellow solid.
-Melting point: about 180-182 ℃.
-Solubility: It is soluble in common organic solvents, such as alcohols, ethers and chlorinated hydrocarbons.
-Stability: relatively stable under conventional experimental conditions.
Use:
5-bromo-1H-indazol-3-ol is a potential pharmaceutical intermediate, commonly used in the preparation of various drugs and biologically active compounds. Specific uses include:
-Drug synthesis: As an intermediate for the synthesis of new drugs, 5-bromo-1H-indazol-3-ol can be used to prepare compounds with antibacterial, antiviral, antitumor or other biological activities.
-Pesticide synthesis: It can also be used for the synthesis of pesticides.
Preparation Method:
5-bromo-1H-indazol-3-ol can be prepared in a variety of ways. The following is one of the common methods:
1. First, 2-nitro-4-methylindazole is reacted with thionyl bromide to obtain 2-bromo-4-methylindazole.
2. 2-bromo-4-methylindazole is then reacted with silver bromide to obtain 5-bromo-4-methylindazole.
3. Finally, 5-bromo-4-methylindazole is reacted with sodium hydroxide to obtain 5-bromo-1H-indazol-3-ol.
Safety Information:
5-bromo-1H-indazol-3-ol is generally relatively safe under correct handling and storage conditions. However, like all chemicals, it should be handled with caution.
-Wear appropriate personal protective equipment such as lab gloves, goggles, and lab coats when performing experimental procedures.
-Avoid inhaling dust or vapors of the compound, using suitable ventilation equipment if necessary.
-Avoid contact with skin or eyes. If there is contact, rinse immediately with plenty of water and seek medical help.
-Follow the correct laboratory procedures and safety guidelines during use.
Nature:
-Appearance: 5-bromo-1H-indazol-3-ol is a white to pale yellow solid.
-Melting point: about 180-182 ℃.
-Solubility: It is soluble in common organic solvents, such as alcohols, ethers and chlorinated hydrocarbons.
-Stability: relatively stable under conventional experimental conditions.
Use:
5-bromo-1H-indazol-3-ol is a potential pharmaceutical intermediate, commonly used in the preparation of various drugs and biologically active compounds. Specific uses include:
-Drug synthesis: As an intermediate for the synthesis of new drugs, 5-bromo-1H-indazol-3-ol can be used to prepare compounds with antibacterial, antiviral, antitumor or other biological activities.
-Pesticide synthesis: It can also be used for the synthesis of pesticides.
Preparation Method:
5-bromo-1H-indazol-3-ol can be prepared in a variety of ways. The following is one of the common methods:
1. First, 2-nitro-4-methylindazole is reacted with thionyl bromide to obtain 2-bromo-4-methylindazole.
2. 2-bromo-4-methylindazole is then reacted with silver bromide to obtain 5-bromo-4-methylindazole.
3. Finally, 5-bromo-4-methylindazole is reacted with sodium hydroxide to obtain 5-bromo-1H-indazol-3-ol.
Safety Information:
5-bromo-1H-indazol-3-ol is generally relatively safe under correct handling and storage conditions. However, like all chemicals, it should be handled with caution.
-Wear appropriate personal protective equipment such as lab gloves, goggles, and lab coats when performing experimental procedures.
-Avoid inhaling dust or vapors of the compound, using suitable ventilation equipment if necessary.
-Avoid contact with skin or eyes. If there is contact, rinse immediately with plenty of water and seek medical help.
-Follow the correct laboratory procedures and safety guidelines during use.
Last Update:2024-04-09 20:45:29
Supplier List
Spot supply
Product Name: 5-Bromo-1H-indazol-3-ol Visit Supplier Webpage Request for quotationCAS: 7364-27-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 5-bromo-1H-indazol-3-ol Request for quotation
CAS: 7364-27-4
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 7364-27-4
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: 5-BROMO-3-HYDROXY (1H)INDAZOLE Request for quotation
CAS: 7364-27-4
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 7364-27-4
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: 5-Bromo-3-Hydroxy (1H)Indazole Visit Supplier Webpage Request for quotation
CAS: 7364-27-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 7364-27-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 5-Bromo-1H-indazol-3-ol Visit Supplier Webpage Request for quotationCAS: 7364-27-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 5-bromo-1H-indazol-3-ol Request for quotation
CAS: 7364-27-4
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 7364-27-4
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: 5-BROMO-3-HYDROXY (1H)INDAZOLE Request for quotation
CAS: 7364-27-4
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 7364-27-4
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: 5-Bromo-3-Hydroxy (1H)Indazole Visit Supplier Webpage Request for quotation
CAS: 7364-27-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 7364-27-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



