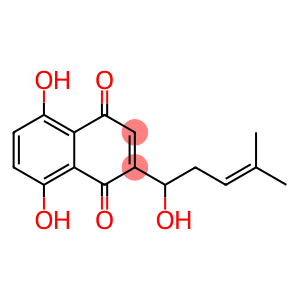
5,8-Dihydroxy-2-(1-hydroxy-4-methylpent-3-enyl)naphthalene-1,4-dione
Shikalkin
CAS: 54952-43-1
Molecular Formula: C16H16O5
5,8-Dihydroxy-2-(1-hydroxy-4-methylpent-3-enyl)naphthalene-1,4-dione - Names and Identifiers
5,8-Dihydroxy-2-(1-hydroxy-4-methylpent-3-enyl)naphthalene-1,4-dione - Physico-chemical Properties
| Molecular Formula | C16H16O5 |
| Molar Mass | 288.3 |
| Density | 1.373±0.06 g/cm3(Predicted) |
| Melting Point | 144-145 °C |
| Boling Point | 567.4±50.0 °C(Predicted) |
| Solubility | methanol: soluble1mg/mL |
| Appearance | powder or solid |
| Color | red to brown |
| pKa | 7.34±0.20(Predicted) |
| Storage Condition | 2-8°C |
| In vitro study | At molar concentrations, shikonin was found to be active in a variety of CC chemokines (CCL2 monocyte chemotactic protein 1), CCL3 (macrophage inflammatory protein 1α),CCL5 (activity-regulating protein, normal T-cell expressed and secreted protein),CXC chemokine (CXCL12 stromal cell-derived factor 1α) as well as the effects of classical chemoattractants inhibit monocyte chemotaxis as well as calcium flux. Shikonin down-regulates macrophage surface expression of CCR5, the major HIV-1 co-receptor. The anti-HIV and anti-inflammatory activities of Shikonin may be related to its interference with the expression and function of chemokine receptors. Shikonin inhibits Calcium-activated chloride channels in intestinal epithelial cells, and this inhibitory effect is achieved in part by inhibiting the activity of potassium channels on the base side. |
| In vivo study | Shikonin significantly delayed intestinal motility in mice and reduced fecal water content in a neonatal mouse model with symptoms of rotavirus diarrhea without affecting the course of viral infection. Shikonin has been reported to have antioxidant, antibacterial, antiparasitic, antiviral and wound healing activities. Shikonin inhibits hypersensitivity and tracheal hyperreactivity in vivo. Intraperitoneal administration of shikonin resulted in some toxic effects, with an LD50 of 20 mg/kg. If administered by oral and intramuscular injection, the absorption rate of shikonin is very fast, and at 1 minute after administration, shikonin can rarely be detected in plasma. The bioavailability of oral shikonin was 34%. |
5,8-Dihydroxy-2-(1-hydroxy-4-methylpent-3-enyl)naphthalene-1,4-dione - Risk and Safety
| Risk Codes | R22 - Harmful if swallowed R50/53 - Very toxic to aquatic organisms, may cause long-term adverse effects in the aquatic environment. |
| Safety Description | 61 - Avoid release to the environment. Refer to special instructions / safety data sheets. |
| UN IDs | UN 3077 9 / PGIII |
| WGK Germany | 3 |
5,8-Dihydroxy-2-(1-hydroxy-4-methylpent-3-enyl)naphthalene-1,4-dione - Reference
| Reference Show more | 1. [IF=4.759] Junjie Feng et al."An integrated data filtering and identification strategy for rapid profiling of chemical constituents, with Arnebiae Radix as an example."J Chromatogr A. 2020 Oct;1629:461496 2. [IF=2.629] Liu Shuang et al."Shikonin Alleviates Endothelial Cell Injury Induced by ox-LDL via AMPK/Nrf2/HO-1 Signaling Pathway."Evid-Based Compl Alt. 2021;2021:5881321 |
5,8-Dihydroxy-2-(1-hydroxy-4-methylpent-3-enyl)naphthalene-1,4-dione - Reference Information
| biological activity | Shikonin (nchusin, archusa acid, Alkanna Red, C. Et al. I. 75535, ISO arnebin 4, NSC 252844), is a potent, specific inhibitor of pyruvent kinase M2 (PKM2). It is the main component of lithospermum, which is a kind of Chinese herbal medicine with a variety of biological activities. Shikonin is also an inhibitor of the TMEM16A chloride channel in the cell fluorescence quenching assay. Shikonin exerts an anti-inflammatory effect by inhibiting tumor necrosis factor-α (TNF-α) and can prevent the activation of the nuclear factor-Δb (NF-Δb) signaling pathway by inhibiting proteasome. |
| Target | Value |
| chemokine receptor () | |
| TNF-α () | |
| Proteasome () | |
| NF-κB () | |
| PKM2 (FBP absence) | 0.3 μM |
Last Update:2024-04-09 02:00:12
Supplier List
Product Name: Shikalkin Request for quotation
CAS: 54952-43-1
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 54952-43-1
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: Shikalkin Request for quotation
CAS: 54952-43-1
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 54952-43-1
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Spot supply
Product Name: Shikalkin Visit Supplier Webpage Request for quotationCAS: 54952-43-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: Shikalkin Request for quotation
CAS: 54952-43-1
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 54952-43-1
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: Shikalkin Request for quotation
CAS: 54952-43-1
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 54952-43-1
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Spot supply
Product Name: Shikalkin Visit Supplier Webpage Request for quotationCAS: 54952-43-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


