4845-58-3
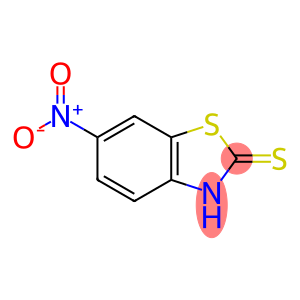
2-Mercapto-6-nitrobenzothiazole
CAS: 4845-58-3
Molecular Formula: C7H4N2O2S2
4845-58-3 - Names and Identifiers
| Name | 2-Mercapto-6-nitrobenzothiazole |
| Synonyms | 2-Benzothiazolethio 2-Benzothiazolethiol, 6-nitro- 2-Mercapto-6-nitrobenzothiazole 6-nitrobenzo[d]thiazole-2-thiol 2-Benzothiazolinethione, 6-nitro- 2-Mercapto-6-nitrobenzo[d]thiazole 2(3H)-Benzothiazolethione, 6-nitro- 6-nitrobenzo[d]thiazole-2(3H)-thione 6-nitro-1,3-benzothiazole-2(3H)-thione 5-nitro-1,3-dihydro-2H-benzimidazole-2-thione |
| CAS | 4845-58-3 |
| EINECS | 678-269-4 |
| InChI | InChI=1/C7H5N3O2S/c11-10(12)4-1-2-5-6(3-4)9-7(13)8-5/h1-3H,(H2,8,9,13) |
4845-58-3 - Physico-chemical Properties
| Molecular Formula | C7H4N2O2S2 |
| Molar Mass | 212.25 |
| Density | 1.5800 (rough estimate) |
| Melting Point | 249-253 °C |
| Boling Point | 389.6±44.0 °C(Predicted) |
| Flash Point | 164.5°C |
| Vapor Presure | 5.03E-05mmHg at 25°C |
| Appearance | Powder |
| Color | Yellow |
| pKa | 7.84±0.20(Predicted) |
| Storage Condition | Sealed in dry,Room Temperature |
| Refractive Index | 1.6000 (estimate) |
4845-58-3 - Risk and Safety
| Hazard Symbols | T - Toxic |
| Risk Codes | R36/37/38 - Irritating to eyes, respiratory system and skin. R25 - Toxic if swallowed R20 - Harmful by inhalation |
| Safety Description | S45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. |
| UN IDs | 2811 |
| RTECS | DL6600000 |
| TSCA | Yes |
| HS Code | 29341000 |
4845-58-3 - Reference Information
| EPA chemical information | Information provided by: ofmpub.epa.gov (external link) |
| There are few reports on the synthesis method of | 2-mercapto-6-nitrobenzothiazole. 2-mercapto-6-nitrobenzothiazole can be prepared by the reaction of 2-chloro-6-nitrobenzothiazole with carbon disulfide; or by the nitration reaction of 2-mercaptobenzothiazole. The reaction formula for the synthesis of 2-mercapto-6-nitrobenzothiazole is shown in the following figure: Experimental operation: add 2-chloro-6-nitrobenzothiazole, carbon disulfide and an appropriate amount of adsorbent to the autoclave as a catalyst, airtight, heating under stirring, and controlling the reaction temperature within the set temperature range. After the reaction is over, cool down, and use 30% sodium hydroxide solution to absorb the hydrogen sulfide gas produced by the reaction. The solid in the kettle is the crude product 2-mercapto-6-nitrobenzothiazole generated by the reaction, and the crude product 2-mercapto-6-nitrobenzothiazole is refined to obtain the finished product. The refining process mainly includes the following steps: sodium hydroxide lye dissolution, filtration, blast oxidation, filtration, sulfuric acid acidification, filtration, washing, vacuum drying, etc. |
Last Update:2024-04-09 19:05:09
Supplier List
Spot supply
Product Name: 2-Mercapto-6-nitrobenzothiazole Visit Supplier Webpage Request for quotationCAS: 4845-58-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-MERCAPTO-6-NITROBENZOTHIAZOLE Request for quotation
CAS: 4845-58-3
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
CAS: 4845-58-3
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
Spot supply
Product Name: 2-Mercapto-6-nitrobenzothiazole Visit Supplier Webpage Request for quotationCAS: 4845-58-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-MERCAPTO-6-NITROBENZOTHIAZOLE Request for quotation
CAS: 4845-58-3
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
CAS: 4845-58-3
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
View History

