475469-12-6
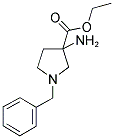
ethyl 3-amino-1-benzylpyrrolidine-3-carboxylate
CAS: 475469-12-6
Molecular Formula: C14H20N2O2
475469-12-6 - Names and Identifiers
475469-12-6 - Physico-chemical Properties
| Molecular Formula | C14H20N2O2 |
| Molar Mass | 248.32 |
| Storage Condition | 2-8°C |
475469-12-6 - Risk and Safety
| Hazard Symbols | Xn - Harmful |
| Risk Codes | 22 - Harmful if swallowed |
475469-12-6 - Introduction
Ethyl is a chemical that can be synthesized in the laboratory. The following are relevant properties, uses, methods and safety information:
Nature:
-Appearance: colorless liquid
-Molecular formula: C15H20N2O2
-Molecular weight: 260.33g/mol
-Melting point: No data
-Boiling Point: No data
-Solubility: Soluble in organic solvents, such as ethanol, dimethylformamide, etc.
Use:
- ethyl l has certain applications in the field of pharmaceutical chemistry. It can be used as a synthetic intermediate for antipsychotic drugs, offering the possibility of synthesizing a wide range of drugs.
Preparation Method:
- ethyl l can be synthesized by a variety of synthetic methods. A common preparation method is to react acetophenone with 3-pyrrolidone to produce 3-benzylpyrrolidine-3-methanone, which is then subjected to reduction and esterification to obtain the target product.
Safety Information:
The toxicological properties and safety data of-ethyl l have not been clearly reported. However, as a compound in organic synthesis, laboratory safety procedures should be followed during use, avoiding contact with skin, eyes and inhalation of gases. In case of accidental contact, rinse immediately with plenty of water and seek medical advice.
Please note that this is only a general introduction to the compounds mentioned, and specific information needs to be further understood and safe operation based on actual use. For chemical experiments and the use of the compound, the laboratory's procedures and instructions should be followed and the relevant laws and regulations should be observed.
Nature:
-Appearance: colorless liquid
-Molecular formula: C15H20N2O2
-Molecular weight: 260.33g/mol
-Melting point: No data
-Boiling Point: No data
-Solubility: Soluble in organic solvents, such as ethanol, dimethylformamide, etc.
Use:
- ethyl l has certain applications in the field of pharmaceutical chemistry. It can be used as a synthetic intermediate for antipsychotic drugs, offering the possibility of synthesizing a wide range of drugs.
Preparation Method:
- ethyl l can be synthesized by a variety of synthetic methods. A common preparation method is to react acetophenone with 3-pyrrolidone to produce 3-benzylpyrrolidine-3-methanone, which is then subjected to reduction and esterification to obtain the target product.
Safety Information:
The toxicological properties and safety data of-ethyl l have not been clearly reported. However, as a compound in organic synthesis, laboratory safety procedures should be followed during use, avoiding contact with skin, eyes and inhalation of gases. In case of accidental contact, rinse immediately with plenty of water and seek medical advice.
Please note that this is only a general introduction to the compounds mentioned, and specific information needs to be further understood and safe operation based on actual use. For chemical experiments and the use of the compound, the laboratory's procedures and instructions should be followed and the relevant laws and regulations should be observed.
Last Update:2024-04-09 20:45:29
Supplier List
Spot supply
Product Name: Ethyl 3-Amino-1-benzyl-3-pyrrolidinecarboxylate Visit Supplier Webpage Request for quotationCAS: 475469-12-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 3-AMINO-1-BENZYL-PYRROLIDINE-3-CARBOXYLIC ACID ETHYL ESTER Request for quotation
CAS: 475469-12-6
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
CAS: 475469-12-6
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
Product Name: 3-AMINO-1-BENZYL-PYRROLIDINE-3-CARBOXYLIC ACID ETHYL ESTER Visit Supplier Webpage Request for quotation
CAS: 475469-12-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 475469-12-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Ethyl 3-Amino-1-benzyl-3-pyrrolidinecarboxylate Visit Supplier Webpage Request for quotationCAS: 475469-12-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 3-AMINO-1-BENZYL-PYRROLIDINE-3-CARBOXYLIC ACID ETHYL ESTER Request for quotation
CAS: 475469-12-6
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
CAS: 475469-12-6
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
Product Name: 3-AMINO-1-BENZYL-PYRROLIDINE-3-CARBOXYLIC ACID ETHYL ESTER Visit Supplier Webpage Request for quotation
CAS: 475469-12-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 475469-12-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


