401815-98-3
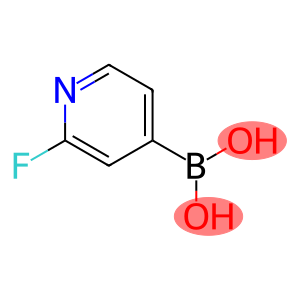
2-Fluoropyridine-4-boronic acid
CAS: 401815-98-3
Molecular Formula: C5H5BFNO2
401815-98-3 - Names and Identifiers
401815-98-3 - Physico-chemical Properties
| Molecular Formula | C5H5BFNO2 |
| Molar Mass | 140.91 |
| Density | 1.34±0.1 g/cm3(Predicted) |
| Melting Point | ca 195℃ |
| Boling Point | 326.8±52.0 °C(Predicted) |
| Flash Point | 151.5°C |
| Solubility | soluble in Methanol |
| Vapor Presure | 8.55E-05mmHg at 25°C |
| Appearance | Solid |
| Color | White to Almost white |
| pKa | 6.51±0.10(Predicted) |
| Storage Condition | Inert atmosphere,Store in freezer, under -20°C |
| Refractive Index | 1.507 |
| MDL | MFCD04112534 |
401815-98-3 - Risk and Safety
| Risk Codes | 36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S37 - Wear suitable gloves. |
| HS Code | 29339900 |
| Hazard Note | Irritant |
401815-98-3 - Reference Information
| Uses | 2-fluoro-4-pyridine boronic acid is a pyridine derivative, which can be used as an intermediate in organic synthesis, and is mainly used for pyridine-containing drug molecules and bioactive molecules Structural modification and derivatization. |
| preparation | preparation steps of 2-fluoro-4-pyridine boronic acid: in the environment of high purity nitrogen, inject 0.0163 mol of 2-fluoro-4-iodopyridine into the experimental reactor, add appropriate amount of tributyl borate and analytically pure tetrahydrofuran, adjust the experimental temperature to 70 ℃, then use a dropping funnel to continue to inject n-butyl lithium solution into the reaction bottle (a slow dropping method should be used in the experiment), and when the reaction solution gradually turns black, continue to maintain a low temperature for 2 h. When the temperature gradually rises to 0 ℃, dilute hydrochloric acid with a mass fraction of 10% (the same below) is added dropwise for hydrolysis, and the pH value of the solution is adjusted to 1.0. Use sodium hydroxide with mass fraction of 25% to adjust the pH value of the solution to 13.0, extract the water-soluble substances in the organic phase, and merge with the separated water phase. Remove organic impurities with n-hexane, and then adjust the pH value of the solution to 9.0 with 10% dilute hydrochloric acid. Add a certain amount of THF to the solution, then add solid sodium chloride to it until the solution reaches saturation, and stir for 1h. Separating again, taking the upper organic phase, taking out the solid under reduced pressure and rotary steaming on a rotary evaporator, purifying and drying to obtain 3.05g product with a yield of 83.11%. |
Last Update:2024-04-09 20:52:54
Supplier List
Spot supply
Product Name: 2-Fluoropyridine-4-boronic acid Visit Supplier Webpage Request for quotationCAS: 401815-98-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-Fluoropyridine-4-boronic acid Request for quotation
CAS: 401815-98-3
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 401815-98-3
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: 2-Fluoropyridine-4-boronic acid Request for quotation
CAS: 401815-98-3
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
CAS: 401815-98-3
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
Product Name: 2-Fluoropyridine-4-boronic acid Visit Supplier Webpage Request for quotation
CAS: 401815-98-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 401815-98-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 2-Fluoropyridine-4-boronic acid Visit Supplier Webpage Request for quotationCAS: 401815-98-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-Fluoropyridine-4-boronic acid Request for quotation
CAS: 401815-98-3
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 401815-98-3
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: 2-Fluoropyridine-4-boronic acid Request for quotation
CAS: 401815-98-3
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
CAS: 401815-98-3
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
Product Name: 2-Fluoropyridine-4-boronic acid Visit Supplier Webpage Request for quotation
CAS: 401815-98-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 401815-98-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



