40059-53-8
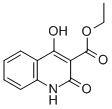
1,2-DIHYDRO-4-HYDROXY-2-OXO-3-QUINOLINECARBOXYLIC ACID ETHYL ESTER
CAS: 40059-53-8
Molecular Formula: C12H11NO4
40059-53-8 - Names and Identifiers
40059-53-8 - Physico-chemical Properties
| Molecular Formula | C12H11NO4 |
| Molar Mass | 233.22 |
| Density | 1.378±0.06 g/cm3(Predicted) |
| Melting Point | 208 °C |
| Boling Point | 458.0±45.0 °C(Predicted) |
| pKa | 4.50±1.00(Predicted) |
| Storage Condition | Sealed in dry,Room Temperature |
40059-53-8 - Upstream Downstream Industry
40059-53-8 - Introduction
ACID ETHYL ESTER is a chemical compound also known as 1,2-dihydro-4-hydroxy-2-keto-3-quinolinecarboxylic ACID ETHYL ESTER. The following is a description of the properties, uses, preparation methods and safety information of the compound:
Nature:
-Appearance: 1, ACID ETHYL ESTER is yellow crystal or powder.
-Molecular formula: C11H9NO4
-Molecular weight: 223.19g/mol
-Melting Point: about 110-112 ° C
-Boiling point: About 370 ° C
Use:
- 1, acethyl ESTER is a drug intermediate commonly used in the synthesis of antibiotics, antiviral drugs and anti-cancer drugs and other drug compounds.
-It can also be used as a photosensitizer, dye and raw material for organic synthesis.
Method:
1. The preparation of ACID ETHYL ESTER can be carried out by the following steps:
1. The amino ketone reacts with formic acid and sulfuric acid to generate 2-hydroxyquinoline -3-carboxylic acid.
2. In anhydrous ether, it reacts with acid chloride to generate 2,4-dihydroxyquinoline -3-carboxylic acid ethyl acetate.
3. Finally, the product is reacted with ethanol to obtain 1, ACID ETHYL ESTER.
Safety Information:
- 1, ACID ETHYL ESTER has certain toxicity and needs to be handled carefully.
-Avoid contact with oxidants, strong bases and strong acids during use or storage.
-Use with suitable protective gloves, glasses and a protective mask to avoid direct contact with skin, eyes and respiratory tract. During operation, good ventilation should be maintained.
-Safety practices should be followed when handling and storing the substance. If you are accidentally poisoned or exposed, you should seek medical attention immediately.
Nature:
-Appearance: 1, ACID ETHYL ESTER is yellow crystal or powder.
-Molecular formula: C11H9NO4
-Molecular weight: 223.19g/mol
-Melting Point: about 110-112 ° C
-Boiling point: About 370 ° C
Use:
- 1, acethyl ESTER is a drug intermediate commonly used in the synthesis of antibiotics, antiviral drugs and anti-cancer drugs and other drug compounds.
-It can also be used as a photosensitizer, dye and raw material for organic synthesis.
Method:
1. The preparation of ACID ETHYL ESTER can be carried out by the following steps:
1. The amino ketone reacts with formic acid and sulfuric acid to generate 2-hydroxyquinoline -3-carboxylic acid.
2. In anhydrous ether, it reacts with acid chloride to generate 2,4-dihydroxyquinoline -3-carboxylic acid ethyl acetate.
3. Finally, the product is reacted with ethanol to obtain 1, ACID ETHYL ESTER.
Safety Information:
- 1, ACID ETHYL ESTER has certain toxicity and needs to be handled carefully.
-Avoid contact with oxidants, strong bases and strong acids during use or storage.
-Use with suitable protective gloves, glasses and a protective mask to avoid direct contact with skin, eyes and respiratory tract. During operation, good ventilation should be maintained.
-Safety practices should be followed when handling and storing the substance. If you are accidentally poisoned or exposed, you should seek medical attention immediately.
Last Update:2024-04-09 21:00:56
Supplier List
Spot supply
Product Name: 1,2-Dihydro-4-hydroxy-2-oxo-3-quinolinecarboxylic acid ethyl ester Visit Supplier Webpage Request for quotationCAS: 40059-53-8
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Product Name: 1,2-DIHYDRO-4-HYDROXY-2-OXO-3-QUINOLINECARBOXYLIC ACID ETHYL ESTER Request for quotation
CAS: 40059-53-8
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
CAS: 40059-53-8
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
Product Name: 1,2-DIHYDRO-4-HYDROXY-2-OXO-3-QUINOLINECARBOXYLIC ACID ETHYL ESTER Visit Supplier Webpage Request for quotation
CAS: 40059-53-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 40059-53-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 1,2-Dihydro-4-hydroxy-2-oxo-3-quinolinecarboxylic acid ethyl ester Visit Supplier Webpage Request for quotationCAS: 40059-53-8
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Product Name: 1,2-DIHYDRO-4-HYDROXY-2-OXO-3-QUINOLINECARBOXYLIC ACID ETHYL ESTER Request for quotation
CAS: 40059-53-8
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
CAS: 40059-53-8
Tel: +86 13001858685
Email: 13001858685@163.com
QQ: 1731087008
WhatsApp: +86 13001858685
Product Name: 1,2-DIHYDRO-4-HYDROXY-2-OXO-3-QUINOLINECARBOXYLIC ACID ETHYL ESTER Visit Supplier Webpage Request for quotation
CAS: 40059-53-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 40059-53-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History
Raw Materials for 40059-53-8
Downstream Products for 40059-53-8


