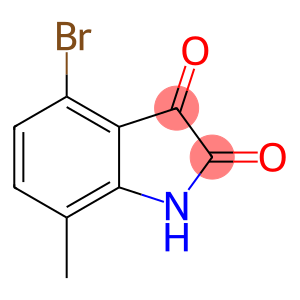
4-bromo-7-methyl-indoline-2,3-dione
4-bromo-7-methyl-indoline-2,3-dione
CAS: 874375-17-4
Molecular Formula: C9H6BrNO2
4-bromo-7-methyl-indoline-2,3-dione - Names and Identifiers
4-bromo-7-methyl-indoline-2,3-dione - Physico-chemical Properties
| Molecular Formula | C9H6BrNO2 |
| Molar Mass | 240.05 |
| Density | 1.714±0.06 g/cm3(Predicted) |
| pKa | 9.56±0.20(Predicted) |
| Storage Condition | 2-8°C |
| Refractive Index | 1.631 |
4-bromo-7-methyl-indoline-2,3-dione - Introduction
4-bromo-7-methyl-indoline-2,3-dione is an organic compound with the chemical formula C9H5BrNO2. It is a red solid crystal that can be dissolved in organic solvents such as ethanol, dimethyl sulfoxide and dichloromethane.
Regarding its properties, it is a compound with strong absorption and emission of red spectrum, so it is often used as a dye in the dye industry. In addition, it also has the antioxidant properties of natural or artificial colors and can be used in antioxidants.
It is widely used in the dye industry and can be used in the synthesis of dyes and pigments, plastic coloring, inks and pigments. It is also used as a colorant and marker in biomedical research.
A common method for preparing 4-bromo-7-methyl-indoline-2,3-dione is to react isatin and bromoethane under alkaline conditions. First, isatin is dissolved in an alkaline solution and then ethyl bromide is added. After completion of the reaction, the product was purified by crystallization to give 4-bromo-7-methyl-indoline-2,3-dione.
Regarding safety information, 4-bromo-7-methyl-indoline-2,3-dione is relatively safe under general operating conditions. However, as an organic compound, it should be handled and stored properly, avoiding contact with the skin and eyes. When using and handling, you need to pay attention to necessary protective measures, such as wearing gloves, goggles and protective clothing. At the same time, it should also be away from fire and oxidizing agents to avoid fire and explosion hazards. Before any use, it is best to carry out a prior safety assessment and laboratory-specified procedures. If ingested or inhaled by mistake, seek medical advice immediately.
Regarding its properties, it is a compound with strong absorption and emission of red spectrum, so it is often used as a dye in the dye industry. In addition, it also has the antioxidant properties of natural or artificial colors and can be used in antioxidants.
It is widely used in the dye industry and can be used in the synthesis of dyes and pigments, plastic coloring, inks and pigments. It is also used as a colorant and marker in biomedical research.
A common method for preparing 4-bromo-7-methyl-indoline-2,3-dione is to react isatin and bromoethane under alkaline conditions. First, isatin is dissolved in an alkaline solution and then ethyl bromide is added. After completion of the reaction, the product was purified by crystallization to give 4-bromo-7-methyl-indoline-2,3-dione.
Regarding safety information, 4-bromo-7-methyl-indoline-2,3-dione is relatively safe under general operating conditions. However, as an organic compound, it should be handled and stored properly, avoiding contact with the skin and eyes. When using and handling, you need to pay attention to necessary protective measures, such as wearing gloves, goggles and protective clothing. At the same time, it should also be away from fire and oxidizing agents to avoid fire and explosion hazards. Before any use, it is best to carry out a prior safety assessment and laboratory-specified procedures. If ingested or inhaled by mistake, seek medical advice immediately.
Last Update:2024-04-10 22:29:15
Supplier List
Product Name: 4-BROMO-7-METHYLISATIN Request for quotation
CAS: 874375-17-4
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 874375-17-4
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: 4-BROMO-7-METHYLISATIN Visit Supplier Webpage Request for quotation
CAS: 874375-17-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 874375-17-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: 4-BROMO-7-METHYLISATIN Request for quotation
CAS: 874375-17-4
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 874375-17-4
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: 4-BROMO-7-METHYLISATIN Visit Supplier Webpage Request for quotation
CAS: 874375-17-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 874375-17-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History

