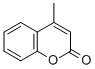
4-Methylcumarin
4-Methylcumarin
CAS: 607-71-6
Molecular Formula: C10H8O2
4-Methylcumarin - Names and Identifiers
4-Methylcumarin - Physico-chemical Properties
| Molecular Formula | C10H8O2 |
| Molar Mass | 160.17 |
| Density | 1.0538 (rough estimate) |
| Melting Point | 79.0 to 83.0 °C |
| Boling Point | 180°C/15mmHg(lit.) |
| Storage Condition | Room Temprature |
| Refractive Index | 1.4108 (estimate) |
| Physical and Chemical Properties | Chemical properties white needle-like or prismatic crystals. The melting point is 83-84 ℃. Soluble in ethanol and benzene. |
| Use | Use as an intermediate in organic synthesis and as a fragrance. |
4-Methylcumarin - Upstream Downstream Industry
| Raw Materials | Ethyl acetoacetate |
4-Methylcumarin - Nature
| storage conditions | Sealed in dry,2-8°C |
| maximum wavelength (& lambda;max) | 270nm(EtOH)(lit.) |
4-Methylcumarin - Production method
It is obtained from the condensation reaction of phenol and ethyl acetoacetate. Stir phenol, ethyl acetoacetate and newly steamed nitrobenzene together, heat to 100 ℃, and add anhydrous aluminum trichloride-nitrobenzene solution dropwise within 45min. The temperature was raised to 130 ℃, and the heat preservation reaction was carried out for 3 hours until hydrogen chloride almost stopped escaping. Cool to room temperature, add hydrochloric acid to decompose excess aluminum trichloride under stirring. Then steam distillation is carried out to remove unreacted ethyl acetoacetate and nitrobenzene. The residue is left to the water layer, the oil layer is filtered and then distilled under reduced pressure. Nitrobenzene is first distilled, and then the fraction at 180-195 ℃(2.0kPa) is collected, which is 4-methylcoumarin.
Supplier List
CAS: 607-71-6
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 607-71-6
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 607-71-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 607-71-6
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 607-71-6
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 607-71-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025


