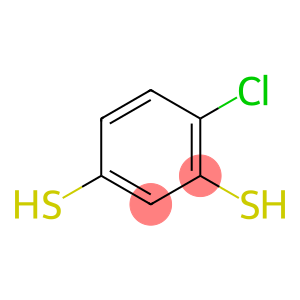
4-Chloro-1,3-benzenedithiole
4-chloro-1,3-benzenedithiol
CAS: 58593-78-5
Molecular Formula: C6H5ClS2
4-Chloro-1,3-benzenedithiole - Names and Identifiers
| Name | 4-chloro-1,3-benzenedithiol |
| Synonyms | 4-Chloro-m-benzenedithiol 4-CHLORO-M-BENZENEDITHIOL 4-Chlorobenzene-1,3-dithiol 4-chlorobenzene-1,3-dithiol 4-chloro-1,3-benzenedithiol 4-CHLORO-1,3-BENZENEDITHIOL 4-Chloro-1,3-benzenedithiole 4-Chlorobenzene-1,3-bisthiol 1,3-Benzenedithiol, 4-chloro- 4-chlorobenzene-1,3-bis(thiolate) |
| CAS | 58593-78-5 |
| EINECS | 261-348-9 |
| InChI | InChI=1/C6H5ClS2/c7-5-2-1-4(8)3-6(5)9/h1-3,8-9H/p-2 |
4-Chloro-1,3-benzenedithiole - Physico-chemical Properties
| Molecular Formula | C6H5ClS2 |
| Molar Mass | 176.69 |
| Density | 1.393g/mLat 25°C(lit.) |
| Melting Point | 31°C |
| Boling Point | 145-146°C/13mmHg(lit.) |
| Flash Point | 113°C |
| Vapor Presure | 0.00488mmHg at 25°C |
| Appearance | solid |
| Refractive Index | n20/D 1.6704(lit.) |
4-Chloro-1,3-benzenedithiole - Risk and Safety
| UN IDs | UN 2810 6.1/PG 3 |
4-Chloro-1,3-benzenedithiole - Introduction
4-chloro-1,3-benzenedithiol, chemical formula C6H4Cl2S2, is an organic sulfur compound. The following is a description of its nature, use, preparation and safety information:
Nature:
- 4-chloro-1,3-benzenedithiol is a white crystalline solid with a peculiar odor of sulfur.
-Its melting point is about 70-76 degrees Celsius, and its boiling point is unknown.
-Soluble in water, soluble in organic solvents such as alcohol and ether.
Use:
- 4-chloro-1,3-benzenedithiol can be used as a reagent in organic synthesis, and is widely used in the extraction and coordination chemical reactions of metal ions.
-It can also be used as a component of photovoltaic materials to enhance the performance of solar cells.
Preparation Method:
- 4-chloro-1,3-benzenedithiol can be prepared by the reaction of benzodithiol and carbon tetrachloride. First, benzodithiol is reacted with sodium bicarbonate under alkaline conditions to produce sodium benzodithiol salt. This sodium salt is then reacted with carbon tetrachloride in the presence of a copper (II) chloride catalyst to give 4-chloro-1,3-benzenedithiol.
Safety Information:
- 4-chloro-1,3-benzenedithiol has strong irritation to eyes, skin and respiratory system, and should be rinsed with plenty of water immediately after contact.
-It has low toxicity, but still requires compliance with laboratory and chemical safety procedures.
-When handling the compound, care should be taken to avoid inhaling its dust or contacting its solution.
Nature:
- 4-chloro-1,3-benzenedithiol is a white crystalline solid with a peculiar odor of sulfur.
-Its melting point is about 70-76 degrees Celsius, and its boiling point is unknown.
-Soluble in water, soluble in organic solvents such as alcohol and ether.
Use:
- 4-chloro-1,3-benzenedithiol can be used as a reagent in organic synthesis, and is widely used in the extraction and coordination chemical reactions of metal ions.
-It can also be used as a component of photovoltaic materials to enhance the performance of solar cells.
Preparation Method:
- 4-chloro-1,3-benzenedithiol can be prepared by the reaction of benzodithiol and carbon tetrachloride. First, benzodithiol is reacted with sodium bicarbonate under alkaline conditions to produce sodium benzodithiol salt. This sodium salt is then reacted with carbon tetrachloride in the presence of a copper (II) chloride catalyst to give 4-chloro-1,3-benzenedithiol.
Safety Information:
- 4-chloro-1,3-benzenedithiol has strong irritation to eyes, skin and respiratory system, and should be rinsed with plenty of water immediately after contact.
-It has low toxicity, but still requires compliance with laboratory and chemical safety procedures.
-When handling the compound, care should be taken to avoid inhaling its dust or contacting its solution.
Last Update:2024-04-10 22:29:15
Supplier List
Product Name: 4-CHLORO-1,3-BENZENEDITHIOL Request for quotation
CAS: 58593-78-5
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 58593-78-5
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: 4-CHLORO-1,3-BENZENEDITHIOL Request for quotation
CAS: 58593-78-5
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 58593-78-5
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
View History
