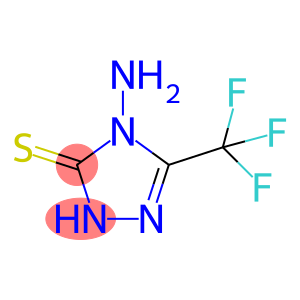
4-AMINO-3-MERCAPTO-5-(TRIFLUOROMETHYL)-4H-1,2,4-TRIAZOLE
4-Amino-5-trifluoromethyl-4H-1,2,4-triazole-3-thiol
CAS: 24848-20-2
Molecular Formula: C3H3F3N4S
4-AMINO-3-MERCAPTO-5-(TRIFLUOROMETHYL)-4H-1,2,4-TRIAZOLE - Names and Identifiers
4-AMINO-3-MERCAPTO-5-(TRIFLUOROMETHYL)-4H-1,2,4-TRIAZOLE - Physico-chemical Properties
| Molecular Formula | C3H3F3N4S |
| Molar Mass | 184.14 |
| Density | 2g/cm3 |
| Boling Point | 140.5°C at 760 mmHg |
| Flash Point | 38.8°C |
| Vapor Presure | 6.12mmHg at 25°C |
| Storage Condition | Room Temprature |
| Refractive Index | 1.642 |
4-AMINO-3-MERCAPTO-5-(TRIFLUOROMETHYL)-4H-1,2,4-TRIAZOLE - Risk and Safety
| Hazard Symbols | Xi - Irritant |
| Hazard Class | IRRITANT |
4-AMINO-3-MERCAPTO-5-(TRIFLUOROMETHYL)-4H-1,2,4-TRIAZOLE - Introduction
-2,4-triazole-3-thiol is an organic compound with the following properties:
1. Appearance: White crystalline solid.
2. Melting point: About 195 ° C.
3. Solubility: soluble in ammonia and some organic solvents, such as ethanol and chloroform.
4. Chemical properties: The compound has the property of ligand sensitive to metal ions, and has certain application value in coordination chemistry and organic synthesis.
The uses of 2,4-triazole-3-thiol mainly include the following aspects:
1. Pesticide: as an intermediate of antifungal pesticide, it is widely used in the prevention and control of rice, fruit trees, vegetables and other crops.
2. Medicine: as an intermediate for the synthesis of anti-tumor, anti-virus and other drugs.
3. Organic Synthesis: used as an important reagent in organic synthesis, can participate in a variety of organic reactions, such as substitution reaction, cyclization reaction.
There are many preparation methods for 2,4-triazole-3-thiol. Commonly used methods include amino reaction and tetrafluoromethanethiol reaction. Generally, trifluoromethyl-4h-1, 2,4-triazole is obtained by reacting an amino compound with sodium carbonyldifluoride, and then the target product is obtained by sulfurization reaction.
It should be noted that 2,4-triazole-3-thiol has certain safety risks, and the following safety operations need to be observed:
1. Avoid inhaling aerosols or dust, and wear respiratory protective equipment if necessary.
2. Avoid skin contact and eye contact, avoid ingestion.
3. Wear appropriate personal protective equipment such as lab gloves, goggles and protective clothing during operation.
4. If you come into contact with this compound, rinse immediately with plenty of water and seek medical attention.
When using and handling this compound, it is important to strictly follow the relevant laboratory safety procedures.
1. Appearance: White crystalline solid.
2. Melting point: About 195 ° C.
3. Solubility: soluble in ammonia and some organic solvents, such as ethanol and chloroform.
4. Chemical properties: The compound has the property of ligand sensitive to metal ions, and has certain application value in coordination chemistry and organic synthesis.
The uses of 2,4-triazole-3-thiol mainly include the following aspects:
1. Pesticide: as an intermediate of antifungal pesticide, it is widely used in the prevention and control of rice, fruit trees, vegetables and other crops.
2. Medicine: as an intermediate for the synthesis of anti-tumor, anti-virus and other drugs.
3. Organic Synthesis: used as an important reagent in organic synthesis, can participate in a variety of organic reactions, such as substitution reaction, cyclization reaction.
There are many preparation methods for 2,4-triazole-3-thiol. Commonly used methods include amino reaction and tetrafluoromethanethiol reaction. Generally, trifluoromethyl-4h-1, 2,4-triazole is obtained by reacting an amino compound with sodium carbonyldifluoride, and then the target product is obtained by sulfurization reaction.
It should be noted that 2,4-triazole-3-thiol has certain safety risks, and the following safety operations need to be observed:
1. Avoid inhaling aerosols or dust, and wear respiratory protective equipment if necessary.
2. Avoid skin contact and eye contact, avoid ingestion.
3. Wear appropriate personal protective equipment such as lab gloves, goggles and protective clothing during operation.
4. If you come into contact with this compound, rinse immediately with plenty of water and seek medical attention.
When using and handling this compound, it is important to strictly follow the relevant laboratory safety procedures.
Last Update:2024-04-10 22:40:09
Supplier List
Product Name: 4-AMINO-5-TRIFLUOROMETHYL-4H-1,2,4-TRIAZOLE-3-THIOL Request for quotation
CAS: 24848-20-2
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 24848-20-2
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Product Name: 4-Amino-5-Trifluoromethyl-4H-1,2,4-Triazole-3-Thiol Visit Supplier Webpage Request for quotation
CAS: 24848-20-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 24848-20-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: 4-AMINO-5-TRIFLUOROMETHYL-4H-1,2,4-TRIAZOLE-3-THIOL Request for quotation
CAS: 24848-20-2
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 24848-20-2
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Product Name: 4-Amino-5-Trifluoromethyl-4H-1,2,4-Triazole-3-Thiol Visit Supplier Webpage Request for quotation
CAS: 24848-20-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 24848-20-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History

