4,6-Dibromopyrimydine
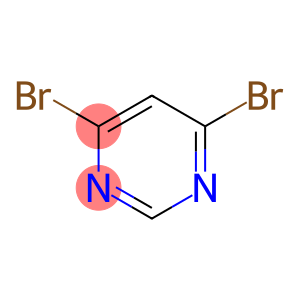
4,6-Dibromopyrimidine
CAS: 36847-10-6
Molecular Formula: C4H2Br2N2
4,6-Dibromopyrimydine - Names and Identifiers
| Name | 4,6-Dibromopyrimidine |
| Synonyms | 4,6-Dibromopyrimydine 4,6-DIBROMOPYRIMIDINE 4,6-Dibromopyrimidine pyrimidine, 4,6-dibromo- Pyrimidine, 4,6-dibromo- |
| CAS | 36847-10-6 |
| InChI | InChI=1/C4H2Br2N2/c5-3-1-4(6)8-2-7-3/h1-2H |
4,6-Dibromopyrimydine - Physico-chemical Properties
| Molecular Formula | C4H2Br2N2 |
| Molar Mass | 237.88 |
| Density | 2?+-.0.06 g/cm3(Predicted) |
| Melting Point | 49-52℃ |
| Boling Point | 263.7±20.0 °C(Predicted) |
| Flash Point | 113.3°C |
| Solubility | soluble in Methanol |
| Vapor Presure | 0.0166mmHg at 25°C |
| Appearance | powder to crystal |
| Color | White to Orange to Green |
| pKa | -4.23±0.17(Predicted) |
| Storage Condition | under inert gas (nitrogen or Argon) at 2-8°C |
| Refractive Index | 1.615 |
4,6-Dibromopyrimydine - Risk and Safety
| UN IDs | UN3261 |
| Hazard Class | 8 |
| Packing Group | III |
4,6-Dibromopyrimydine - Introduction
4,6-Dibromopyrimidine(4,6-Dibromopyrimidine) is an organic compound with the chemical formula C4H2Br2N2. The following is a description of its nature, use, formulation and safety information:
Nature:
-Appearance: 4,6-Dibromopyrimidine is a white to pale yellow crystalline solid.
-Solubility: It is soluble in most organic solvents, such as ethanol, dimethylformamide and chloroform.
-Melting point: The melting point of 4,6-Dibromopyrimidine is about 114-115 degrees Celsius.
-Density: Its density is about 2.25g/cm³.
Use:
-Chemical synthesis intermediate: 4,6-Dibromopyrimidine can be used to prepare other compounds, such as pesticides, drugs and organic synthesis intermediates.
-Pyrimidine acid dyes: It can also be used to prepare pyrimidine acid dyes for applications such as dyes, printing inks and coatings.
Preparation Method:
The preparation method of 4,6-Dibromopyrimidine usually involves the following steps:
1. In the presence of a base, 2,4, 6-tribromopyrimidine reacts with 2,4, 6-trinitropyrimidine to generate 4,6-dinitropyrimidine.
2.4,6-dinitropyrimidine is refluxed with zinc powder, and 4,6-diiodopyrimidine is generated by hydrogenation and denitration reaction.
3. Finally, 4,6-diiodopyrimidine is reacted with sulfoxide to generate 4,6-Dibromopyrimidine.
Safety Information:
- 4,6-Dibromopyrimidine may cause irritation when in contact with skin, eyes or inhalation, and direct contact with it should be avoided.
-Wear appropriate protective gloves, goggles and respiratory protection when handling the compound.
-Because it may be toxic to aquatic life, avoid discharging it into water bodies.
-Please use, handle and store the compound properly under the guidance of a professional.
Nature:
-Appearance: 4,6-Dibromopyrimidine is a white to pale yellow crystalline solid.
-Solubility: It is soluble in most organic solvents, such as ethanol, dimethylformamide and chloroform.
-Melting point: The melting point of 4,6-Dibromopyrimidine is about 114-115 degrees Celsius.
-Density: Its density is about 2.25g/cm³.
Use:
-Chemical synthesis intermediate: 4,6-Dibromopyrimidine can be used to prepare other compounds, such as pesticides, drugs and organic synthesis intermediates.
-Pyrimidine acid dyes: It can also be used to prepare pyrimidine acid dyes for applications such as dyes, printing inks and coatings.
Preparation Method:
The preparation method of 4,6-Dibromopyrimidine usually involves the following steps:
1. In the presence of a base, 2,4, 6-tribromopyrimidine reacts with 2,4, 6-trinitropyrimidine to generate 4,6-dinitropyrimidine.
2.4,6-dinitropyrimidine is refluxed with zinc powder, and 4,6-diiodopyrimidine is generated by hydrogenation and denitration reaction.
3. Finally, 4,6-diiodopyrimidine is reacted with sulfoxide to generate 4,6-Dibromopyrimidine.
Safety Information:
- 4,6-Dibromopyrimidine may cause irritation when in contact with skin, eyes or inhalation, and direct contact with it should be avoided.
-Wear appropriate protective gloves, goggles and respiratory protection when handling the compound.
-Because it may be toxic to aquatic life, avoid discharging it into water bodies.
-Please use, handle and store the compound properly under the guidance of a professional.
Last Update:2024-04-09 21:04:16
Supplier List
Spot supply
Product Name: 4,6-Dibromopyrimidine, 95% Visit Supplier Webpage Request for quotationCAS: 36847-10-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 4,6-DIBROMOPYRIMIDINE Request for quotation
CAS: 36847-10-6
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 36847-10-6
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: 4,6-Dibromopyrimidine Visit Supplier Webpage Request for quotation
CAS: 36847-10-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 36847-10-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 4,6-Dibromopyrimidine, 95% Visit Supplier Webpage Request for quotationCAS: 36847-10-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 4,6-DIBROMOPYRIMIDINE Request for quotation
CAS: 36847-10-6
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 36847-10-6
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: 4,6-Dibromopyrimidine Visit Supplier Webpage Request for quotation
CAS: 36847-10-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 36847-10-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


