3209-70-9
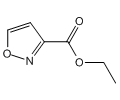
3-Isoxazolecarboxylicacid, ethyl ester
CAS: 3209-70-9
Molecular Formula: C6H7NO3
3209-70-9 - Names and Identifiers
3209-70-9 - Physico-chemical Properties
| Molecular Formula | C6H7NO3 |
| Molar Mass | 141.12 |
| Density | 1.177 |
| Boling Point | 219℃ |
| Flash Point | 87℃ |
| pKa | -5.43±0.50(Predicted) |
| Storage Condition | Sealed in dry,Store in freezer, under -20°C |
3209-70-9 - Reference Information
| Use | Ethyl 3-isoxazole carboxylic acid is used as a reactant to prepare 2-amino -3-(3-carboxy-5-methyl-4-isoxazole) propionic acid (ACPA),ACPA is active on metabolic receptors and is a weak antagonist of mGlu2 receptor subtypes. Isoxazole, as a ubiquitous heterocyclic dominant structural skeleton, has shown a wide range of pharmacological activities, such as anti-tumor, antibacterial, anti-diabetic, antiviral, acaricidal, etc. Therefore, it has attracted extensive attention of many pharmaceutical chemists. |
| preparation | traditional methods for synthesizing isoxazole heterocycles mainly include: hydroxyamine compounds undergo [3+2] cycloaddition reaction with alkynes after N-chlorinated succinimide (NCS) chlorination; After oxidation, nitrile compounds are subjected to 1,3-Dipolar cycloaddition reaction. Although the above method has made a great contribution to the construction of functional isoxazole compounds, there are many by-products, low yield and other shortcomings, especially can not be directly applied to the synthesis of polyphenol hydroxyphenyl substituted isoxazole. In this paper, acetophenone is used as the starting material, sodium hydride is used as a strong base and diethyl oxalate is condensed to obtain enol-type intermediates, and then directly condensed with hydroxylamine hydrochloride to obtain ethyl 3-isoxazole carboxylate. The synthesis reaction formula is as follows: |
Last Update:2024-04-09 21:54:55
Supplier List
Spot supply
Product Name: ETHYL ISOXAZOLE-3-CARBOXYLATE Visit Supplier Webpage Request for quotationCAS: 3209-70-9
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: ETHYL ISOXAZOLE-3-CARBOXYLATE Request for quotation
CAS: 3209-70-9
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 3209-70-9
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: Ethyl Isoxazol-3-carboxylate Visit Supplier Webpage Request for quotation
CAS: 3209-70-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 3209-70-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: ETHYL ISOXAZOLE-3-CARBOXYLATE Visit Supplier Webpage Request for quotationCAS: 3209-70-9
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: ETHYL ISOXAZOLE-3-CARBOXYLATE Request for quotation
CAS: 3209-70-9
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 3209-70-9
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: Ethyl Isoxazol-3-carboxylate Visit Supplier Webpage Request for quotation
CAS: 3209-70-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 3209-70-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


