30558-43-1
Glyphosate
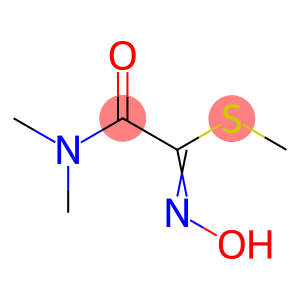
CAS: 30558-43-1
Molecular Formula: C5H10N2O2S
30558-43-1 - Names and Identifiers
30558-43-1 - Physico-chemical Properties
| Molecular Formula | C5H10N2O2S |
| Molar Mass | 162.21 |
| Density | 1.213 (estimate) |
| Boling Point | 272.1±23.0 °C(Predicted) |
| pKa | 9.39±0.10(Predicted) |
| Storage Condition | 2-8℃ |
| Refractive Index | 1.6430 (estimate) |
30558-43-1 - Risk and Safety
| Toxicity | LD50 oral in rat: > 7gm/kg |
30558-43-1 - Introduction
Oximes are a class of organic compounds that contain a carbonyl group (C=O) linked to an imino group (-NH-) by a carbon atom. Here is some information about the properties, uses, preparation methods, and safety considerations of oximes:
Properties:
- Oximes have a characteristic oxime functional group (-C=NOH) and can exist in either liquid or solid form.
- They are generally polar compounds and have higher boiling points compared to similar hydrocarbons.
- Oximes are usually colorless or light-colored compounds with a distinct odor.
- They can form hydrogen bonds, making them soluble in polar solvents.
Uses:
- Oximes have applications in various fields, including pharmaceuticals, organic synthesis, and chemical analysis.
- One significant application of oximes is in the reactivation of organophosphate-inhibited cholinesterases in cases of poisoning.
- They are used as intermediates in the synthesis of pharmaceuticals, pesticides, and rubber antioxidants.
- Oximes are also employed as catalysts, complexing agents, and stabilizers in chemical reactions.
Preparation methods:
- Oximes can be prepared by the reaction between an aldehyde or ketone with hydroxylamine hydrochloride in the presence of a base, such as sodium hydroxide.
- Another method involves the reaction of a nitrite salt with a primary amine in an acidic or basic medium.
- In some cases, oximes can also be obtained via the reaction of nitriles with hydroxylamine and a suitable catalyst.
Safety information:
- The toxicity of oximes can vary depending on the specific compound. Some oximes may be irritants or sensitizers.
- It is important to handle oximes with caution, including the use of appropriate personal protective equipment (gloves, goggles, lab coat) to prevent direct contact with the skin, eyes, or inhalation of the compounds.
- Proper storage and handling practices should be followed, such as storing oximes in tightly sealed containers in a cool, dry, and well-ventilated area.
- Always consult safety data sheets (SDS) and follow the recommended guidelines provided by the manufacturer or regulatory agencies.
- Disposal of oxime compounds should be carried out in accordance with local regulations and in an environmentally responsible manner.
As always, it is crucial to refer to specific scientific literature, material safety data sheets, and expert guidance for comprehensive information regarding the properties, uses, preparation, and safety considerations of individual oxime compounds.
Supplier List
CAS: 30558-43-1
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
CAS: 30558-43-1
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 30558-43-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 30558-43-1
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
CAS: 30558-43-1
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 30558-43-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025


