3-anilino-1-pyrrolidinecarboxamide
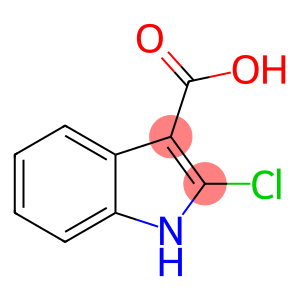
2-chloro-1H-indole-3-carboxylic acid
CAS: 54778-20-0
Molecular Formula: C9H6ClNO2
3-anilino-1-pyrrolidinecarboxamide - Names and Identifiers
3-anilino-1-pyrrolidinecarboxamide - Physico-chemical Properties
| Molecular Formula | C9H6ClNO2 |
| Molar Mass | 195.6 |
| Density | 1.548g/cm3 |
| Boling Point | 434.4°C at 760 mmHg |
| Flash Point | 216.5°C |
| Vapor Presure | 2.56E-08mmHg at 25°C |
| Storage Condition | 2-8°C |
| Refractive Index | 1.728 |
3-anilino-1-pyrrolidinecarboxamide - Introduction
Acid is an organic compound with the chemical formula C9H6ClNO2. The following is a description of its nature, use, preparation and safety information:
Nature:
-Appearance: acid is a white crystalline solid.
-Solubility: Soluble in some organic solvents, such as chloroform, acetone and dichloromethane, slightly soluble in water.
-Melting point: about 189-192°C.
Use:
-Compounds have potential biological activities in the pharmaceutical field, so they may be used as intermediates for pharmaceutical research and development.
-must acid can also be used as a reaction reagent in organic synthesis.
Preparation Method:
-the preparation of acid can be carried out by a variety of methods, such:
-React 1H-indigo-3-carboxylic acid with cuprous hydroxide under alkaline conditions, and then add metal chloride or methyl chloride.
-The substrate 3-carboxylic acid is mixed with sulfuric acid, chlorine and nitrous acid, and then cuprous oxide is oxidized to produce indigo -1H.
Safety Information:
-the safety of acid has not been fully evaluated.
-Good laboratory practices should be followed during use.
-Investigators should use appropriate personal protective equipment such as lab coats, gloves, and goggles.
-When disposing of waste, comply with local waste management regulations.
Nature:
-Appearance: acid is a white crystalline solid.
-Solubility: Soluble in some organic solvents, such as chloroform, acetone and dichloromethane, slightly soluble in water.
-Melting point: about 189-192°C.
Use:
-Compounds have potential biological activities in the pharmaceutical field, so they may be used as intermediates for pharmaceutical research and development.
-must acid can also be used as a reaction reagent in organic synthesis.
Preparation Method:
-the preparation of acid can be carried out by a variety of methods, such:
-React 1H-indigo-3-carboxylic acid with cuprous hydroxide under alkaline conditions, and then add metal chloride or methyl chloride.
-The substrate 3-carboxylic acid is mixed with sulfuric acid, chlorine and nitrous acid, and then cuprous oxide is oxidized to produce indigo -1H.
Safety Information:
-the safety of acid has not been fully evaluated.
-Good laboratory practices should be followed during use.
-Investigators should use appropriate personal protective equipment such as lab coats, gloves, and goggles.
-When disposing of waste, comply with local waste management regulations.
Last Update:2024-04-09 02:00:46
Supplier List
Spot supply
Product Name: 2-CHLORO-1H-INDOLE-3-CARBOXYLIC ACID Visit Supplier Webpage Request for quotationCAS: 54778-20-0
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-CHLORO-1H-INDOLE-3-CARBOXYLIC ACID Request for quotation
CAS: 54778-20-0
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 54778-20-0
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: 2-CHLORO-1H-INDOLE-3-CARBOXYLIC ACID Visit Supplier Webpage Request for quotation
CAS: 54778-20-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 54778-20-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 2-CHLORO-1H-INDOLE-3-CARBOXYLIC ACID Visit Supplier Webpage Request for quotationCAS: 54778-20-0
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-CHLORO-1H-INDOLE-3-CARBOXYLIC ACID Request for quotation
CAS: 54778-20-0
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 54778-20-0
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: 2-CHLORO-1H-INDOLE-3-CARBOXYLIC ACID Visit Supplier Webpage Request for quotation
CAS: 54778-20-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 54778-20-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


