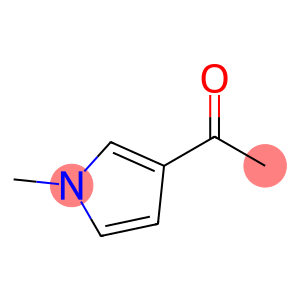
3-acetyl-1-methylpyrrole
3-acetyl-1-methylpyrrole
CAS: 932-62-7
Molecular Formula: C7H9NO
3-acetyl-1-methylpyrrole - Names and Identifiers
| Name | 3-acetyl-1-methylpyrrole |
| Synonyms | Acetylmethylpyrrole 3-acetyl-1-methylpyrrole 3-ACETYL-1-METHYLPYRROLE 1-Methyl-3-acetyl-1H-pyrrole 3-Acetyl-1-methyl-1H-pyrrole 1-(1-methyl-pyrrol-3-yl)-ethanone METHYL 1-METHYL-3-PYRROLYL KETONE 1-(1-Methyl-1H-pyrrol-3-yl)ethanone 1-(1-methyl-1H-pyrrol-3-yl)ethanone 1-(1-Methyl-1H-pyrrol-3-yl)-ethanone |
| CAS | 932-62-7 |
| EINECS | 604-604-1 |
| InChI | InChI=1/C7H9NO/c1-6(9)7-3-4-8(2)5-7/h3-5H,1-2H3 |
3-acetyl-1-methylpyrrole - Physico-chemical Properties
| Molecular Formula | C7H9NO |
| Molar Mass | 123.15 |
| Density | 1.038g/mLat 25°C(lit.) |
| Boling Point | 148-150°C15mm Hg(lit.) |
| Flash Point | >230°F |
| Vapor Presure | 0.292mmHg at 25°C |
| Specific Gravity | 1.061.038 |
| pKa | -5.84±0.70(Predicted) |
| Storage Condition | Room Temprature |
| Refractive Index | n20/D 1.538(lit.) |
| MDL | MFCD00011563 |
3-acetyl-1-methylpyrrole - Risk and Safety
| Hazard Symbols | Xi - Irritant |
| Risk Codes | 36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S37/39 - Wear suitable gloves and eye/face protection |
| WGK Germany | 3 |
3-acetyl-1-methylpyrrole - Reference Information
| Introduction | 3-acetyl-1-methylpyrrole belongs to heterocyclic compounds containing 3-acylpyrrole fragments. Such compounds are useful for the preparation of novel pharmacologically active molecules. For example, 1-alkyl-3-(naphthoyl) pyrrole and 1-alkyl-3-(naphthoyl) indole, which are known to have cannabinoid activity, and 3-acetyl -4-hydroxy methyl pyrrole with antibiotic activity isolated from wart fruit black skin pathogen. |
| preparation | to a stirred solution containing tetrabutylammonium bromide (0.3g,1 mmol) and K2CO3(1.6g,12 mmol) to a suspension of 2-butanone (25 mL) was added 3-acetylpyrrole (1.1g,10 mmol) and methyl bromide (11mmol). The mixture was heated at 60 °c for 12 h and then the reaction was cooled to room temperature. The solvent was removed under reduced pressure. The residue was taken up in H2O/Et2O (1:1, 50ml), the organic layer separated, dried (Na2SO4) and evaporated to give 3-acetyl-1-methylpyrrole. |
Last Update:2024-04-09 02:00:12
Supplier List
Product Name: 3-acetyl-1-methylpyrrole Request for quotation
CAS: 932-62-7
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 932-62-7
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: 3-ACETYL-1-METHYLPYRROLE Request for quotation
CAS: 932-62-7
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 932-62-7
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Spot supply
Product Name: 3-Acetyl-1-Methylpyrrole Visit Supplier Webpage Request for quotationCAS: 932-62-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: 3-acetyl-1-methylpyrrole Request for quotation
CAS: 932-62-7
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 932-62-7
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: 3-ACETYL-1-METHYLPYRROLE Request for quotation
CAS: 932-62-7
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 932-62-7
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Spot supply
Product Name: 3-Acetyl-1-Methylpyrrole Visit Supplier Webpage Request for quotationCAS: 932-62-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


