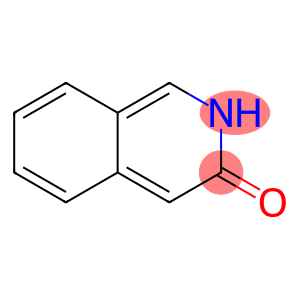
3-ISOQUINOLINOL
3-hydroxyisoquinoline
CAS: 7651-81-2
Molecular Formula: C9H7NO
3-ISOQUINOLINOL - Names and Identifiers
| Name | 3-hydroxyisoquinoline |
| Synonyms | 3-Isoquinolinol 3-ISOQUINOLINOL Isoquinolin-3-ol isoquinolin-3-ol 3(2H)-Isoquinolinone isoquinolin-3(2H)-one Isoquinolin-3(2H)-one 3-HYDROXYISOQUINOLINE 3-Hydorxyisoquinoline 3-hydroxyisoquinoline 2,3-dihydroisoquinolin-3-one Isoquinolin-3-ol, 3-Hydroxy-2-azanaphthalene |
| CAS | 7651-81-2 |
| InChI | InChI=1/C9H7NO/c11-9-5-7-3-1-2-4-8(7)6-10-9/h1-6H,(H,10,11) |
3-ISOQUINOLINOL - Physico-chemical Properties
| Molecular Formula | C9H7NO |
| Molar Mass | 145.16 |
| Density | 1.23±0.1 g/cm3(Predicted) |
| Melting Point | 192-194 °C (lit.) |
| Boling Point | 551.9±23.0 °C(Predicted) |
| Flash Point | 319.4°C |
| Vapor Presure | 3.16E-12mmHg at 25°C |
| BRN | 113230 |
| pKa | 11.06±0.20(Predicted) |
| Storage Condition | Inert atmosphere,Room Temperature |
| Refractive Index | 1.642 |
| MDL | MFCD00075524 |
3-ISOQUINOLINOL - Risk and Safety
| Risk Codes | R22 - Harmful if swallowed R41 - Risk of serious damage to eyes |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S37/39 - Wear suitable gloves and eye/face protection |
| WGK Germany | 3 |
| HS Code | 29334900 |
| Hazard Note | Irritant |
3-ISOQUINOLINOL - Reference Information
| preparation | The resealable reaction tube containing the magnetic follower was sealed with a rubber spacer. The tube was dried with a flame under a flow of argon. tBu3P. HBF4(10.9 mg, 0.0377 mmol) and Pd2(dba)3(17.3 mg, 0.0189 mmol) were added to the tube. The aryl bromide (83.6 mg) was dissolved in anhydrous toluene (0.9 ml) . The resulting solution was added to the tube via syringe. Tert-butyl acetate (87.6 mg) was added to the tube. The reaction mixture was cooled to -78°C. The reaction mixture was degassed with argon for 15 min. Degassed LiHDMS solution (0.942 ml, 1m in toluene) was added to the mixture via syringe. The reaction mixture was degassed for 15 min. Replace the rubber diaphragm with a screw cap. The reaction was stirred at room temperature for 16 hours. The reaction was quenched by the addition of saturated aqueous NaHCO3 (25 mL). The aqueous layer was extracted with Et2O (3 x 25 mL). The combined organics were dried over Na2SO4. The combined organics were filtered. The solvent was removed in vacuo. The product was purified by flash column chromatography [petroleum/EtOAc 50:1, fractionated to 5:1]. |
| Use | 3-hydroxyisoquinoline is a compound used in a polymer mixture of polyvinyl alcohol (PVA) and Nile red, white light can be generated by Forster resonance energy transfer. Can be used as organic, pharmaceutical intermediates. |
Last Update:2024-04-09 15:16:52
Supplier List
Spot supply
Product Name: 3-Hydroxyisoquinoline Visit Supplier Webpage Request for quotationCAS: 7651-81-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 3-hydroxyisoquinoline Request for quotation
CAS: 7651-81-2
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 7651-81-2
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Multiple Specifications
Product Name: 3-Hydroxyisoquinoline Request for quotationCAS: 7651-81-2
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: 3-Hydroxyisoquinoline Visit Supplier Webpage Request for quotation
CAS: 7651-81-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 7651-81-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 3-Hydroxyisoquinoline Visit Supplier Webpage Request for quotationCAS: 7651-81-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 3-hydroxyisoquinoline Request for quotation
CAS: 7651-81-2
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 7651-81-2
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Multiple Specifications
Product Name: 3-Hydroxyisoquinoline Request for quotationCAS: 7651-81-2
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: 3-Hydroxyisoquinoline Visit Supplier Webpage Request for quotation
CAS: 7651-81-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 7651-81-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



