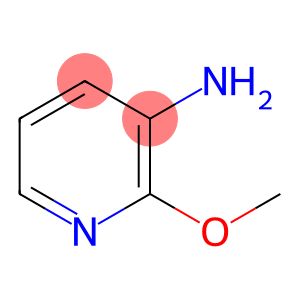
3-Amino-2-methoxypyridine
2-Methoxy-3-aminopyridine
CAS: 20265-38-7
Molecular Formula: C6H8N2O
3-Amino-2-methoxypyridine - Names and Identifiers
| Name | 2-Methoxy-3-aminopyridine |
| Synonyms | TIMTEC-BB SBB005483 2-methoxypyridin-3-amine 2-METHOXYPYRIDIN-3-AMINE 2-Methoxypyridine-3-amine 3-AMINO-2-METHOXYPYRIDINE 3-Amino-2-methoxypyridine 2-Methoxy-3-aminopyridine 2-METHOXY-PYRIDIN-3-YLAMINE |
| CAS | 20265-38-7 |
| EINECS | 673-107-9 |
| InChI | InChI=1/C6H8N2O/c1-9-6-5(7)3-2-4-8-6/h2-4H,7H2,1H3 |
3-Amino-2-methoxypyridine - Physico-chemical Properties
| Molecular Formula | C6H8N2O |
| Molar Mass | 124.14 |
| Density | 1.139±0.06 g/cm3(Predicted) |
| Melting Point | 67 °C |
| Boling Point | 118°C/13mmHg(lit.) |
| Flash Point | 99.3°C |
| Solubility | soluble in Methanol |
| Vapor Presure | 0.0377mmHg at 25°C |
| Appearance | Solid |
| Color | Orange to Brown to Dark red |
| pKa | 4.33±0.10(Predicted) |
| Storage Condition | Keep in dark place,Inert atmosphere,Room temperature |
| Refractive Index | 1.56 |
| MDL | MFCD00833386 |
3-Amino-2-methoxypyridine - Risk and Safety
| Risk Codes | R20/21/22 - Harmful by inhalation, in contact with skin and if swallowed. R36/37/38 - Irritating to eyes, respiratory system and skin. R41 - Risk of serious damage to eyes R37/38 - Irritating to respiratory system and skin. R22 - Harmful if swallowed |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. S39 - Wear eye / face protection. |
| HS Code | 29339900 |
| Hazard Class | IRRITANT |
3-Amino-2-methoxypyridine - Reference Information
| introduction | 2-methoxy-3-aminopyridine is crystalline powder at normal temperature and pressure. it has strong alkalinity and is a commonly used organic heterocyclic block. it can be used to make vitamins, sulfonamides, pesticides and plastics. |
| Use | 2-methoxy-3-aminopyridine can be used as an intermediate in pharmaceutical chemistry and organic synthesis. In the synthetic transformation, the amino group on the pyridine ring can be diazotized and then subjected to subsequent derivatization such as in-situ reduction into hydrazine compounds; in addition. Under appropriate halogenation reaction conditions, a halogen atom can be introduced at the fourth position of pyridine. |
| preparation method | nitro precursor compound (20.8 g, 135 mmol) is dissolved in methanol (270 mL) and palladium carbon (10% Pd dry weight, Degussa E101 NE/W, 50% water content, 5.75g, 2.7 mmol Pd) is added, displace the air in the reaction bottle with hydrogen (switch between vacuum and hydrogen from the balloon five times), stir the mixture overnight, and then filter. The filtrate is vacuum concentrated, the residue is dissolved in a 1:1 hexane: ethyl acetate mixture, and washed with water and saturated sodium bicarbonate to separate the organic layer, the organic layer is dried with anhydrous magnesium sulfate, and filtered to remove the magnesium sulfate precipitate. The filtrate can be concentrated under reduced pressure to obtain the target product 2-methoxy-3-aminopyridine (12.43 g, 74%), which is a white solid. Fig. 2-methoxy-3-aminopyridine synthesis route |
Last Update:2024-04-09 21:11:58
Supplier List
Spot supply
Product Name: 3-Amino-2-methoxypyridine Visit Supplier Webpage Request for quotationCAS: 20265-38-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-Methoxy-3-aminopyridine Request for quotation
CAS: 20265-38-7
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 20265-38-7
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Spot supply
Product Name: 2-Methoxypyridin-3-amine Visit Supplier Webpage Request for quotationCAS: 20265-38-7
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: 2-Methoxypyridin-3-amine Request for quotationCAS: 20265-38-7
Tel: 15335994747
Email: 3521670276@qq.com
Mobile: 15335994747
QQ: 3521670276
Wechat: A2020071098
Product Name: 2-Methoxypyridin-3-amine Request for quotation
CAS: 20265-38-7
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 20265-38-7
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Product Name: 2-Methoxypyridin-3-amine Request for quotation
CAS: 20265-38-7
Tel: 0086-13605431940
Email: xuli678323@126.com
Mobile: +86-13605431940
QQ: 56860987
Wechat: 13605431940
WhatsApp: 13605431940
CAS: 20265-38-7
Tel: 0086-13605431940
Email: xuli678323@126.com
Mobile: +86-13605431940
QQ: 56860987
Wechat: 13605431940
WhatsApp: 13605431940
Product Name: 2-Methoxypyridin-3-amine Visit Supplier Webpage Request for quotation
CAS: 20265-38-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 20265-38-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 3-Amino-2-methoxypyridine Visit Supplier Webpage Request for quotationCAS: 20265-38-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-Methoxy-3-aminopyridine Request for quotation
CAS: 20265-38-7
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 20265-38-7
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Spot supply
Product Name: 2-Methoxypyridin-3-amine Visit Supplier Webpage Request for quotationCAS: 20265-38-7
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: 2-Methoxypyridin-3-amine Request for quotationCAS: 20265-38-7
Tel: 15335994747
Email: 3521670276@qq.com
Mobile: 15335994747
QQ: 3521670276
Wechat: A2020071098
Product Name: 2-Methoxypyridin-3-amine Request for quotation
CAS: 20265-38-7
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 20265-38-7
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Product Name: 2-Methoxypyridin-3-amine Request for quotation
CAS: 20265-38-7
Tel: 0086-13605431940
Email: xuli678323@126.com
Mobile: +86-13605431940
QQ: 56860987
Wechat: 13605431940
WhatsApp: 13605431940
CAS: 20265-38-7
Tel: 0086-13605431940
Email: xuli678323@126.com
Mobile: +86-13605431940
QQ: 56860987
Wechat: 13605431940
WhatsApp: 13605431940
Product Name: 2-Methoxypyridin-3-amine Visit Supplier Webpage Request for quotation
CAS: 20265-38-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 20265-38-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History






