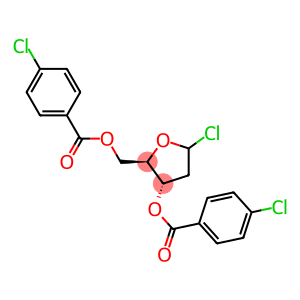
3,5-O-Bis(4-chlorobenzoyl)-2-deoxy-D-ribofuranosyl
1-Chloro-3,5-di(4-chlorbenzoyl)-2-deoxy-D-ribose
CAS: 3601-90-9
Molecular Formula: C19H15Cl3O5
3,5-O-Bis(4-chlorobenzoyl)-2-deoxy-D-ribofuranosyl - Names and Identifiers
| Name | 1-Chloro-3,5-di(4-chlorbenzoyl)-2-deoxy-D-ribose |
| Synonyms | 1-Chloro-3,5-Di-(P-Chlorobenzo 1-CHLORO-3,5-DI(4-CHLORBENZOYL)-2-DEOXY-D-RIBOSE 1-Chloro-3,5-di(4-chlorbenzoyl)-2-deoxy-D-ribose 1-CHLORO-3,5-DIPARACHLOROBENZOYL-2-DEOXY-D-RIBOSE 1-Chloro-3,5-di-(p-chlorobenzoyl)-2-deoxy-D-ribose 2-Deoxy-3,5-bis-O-(4-chlorobenzoyl)-D-erythro-pent 3,5-O-Bis(4-chlorobenzoyl)-2-deoxy-D-ribofuranosyl 1-Chloro-3,5-di-(p-chlorobenzoyl)-2-deoxy-D-ribofuranose 1-Chloro-2-Deoxy-3,5-Di(O-P-Chlorobenzoyl)-D-Ribofuranose 1-Chloro-2-deoxy-3,5-bi (O-p-aldylbenzoyl)-D-ribofuranose 3,5-O-Bis(4-chlorobenzoyl)-2-deoxy-D-ribofuranosyl chloride |
| CAS | 3601-90-9 |
| EINECS | 1806241-263-5 |
| InChI | InChI=1/C19H15Cl3O6/c20-12-5-1-10(2-6-12)15(24)16(25)18(27)19(28,9-14(22)23)17(26)11-3-7-13(21)8-4-11/h1-8,16,18,25,27-28H,9H2/t16?,18-,19-/m1/s1 |
3,5-O-Bis(4-chlorobenzoyl)-2-deoxy-D-ribofuranosyl - Physico-chemical Properties
| Molecular Formula | C19H15Cl3O5 |
| Molar Mass | 429.68 |
| Density | 1.46 |
| Boling Point | 530.6±50.0 °C(Predicted) |
| Flash Point | 366.4°C |
| Vapor Presure | 1.5E-19mmHg at 25°C |
| Storage Condition | 2-8°C |
| Refractive Index | 1.636 |
3,5-O-Bis(4-chlorobenzoyl)-2-deoxy-D-ribofuranosyl - Reference Information
| Properties | 1-chloro-3, 5-di-p-chlorobenzoyloxy-2-deoxy-D-ribose is white at room temperature and pressure Solid. |
| Use | 1-chloro-3, 5-di-p-chlorobenzoyloxy-2-deoxy-D-ribose is an intermediate in pharmaceutical chemistry and organic synthesis, For example, it can be used for the preparation and synthesis of the drug molecule decitabine, which is a natural adenosine analog of 2 '-deoxycytidine, by inhibiting DNA methyltransferase, reducing DNA methylation, thereby inhibiting tumor cell proliferation and preventing the occurrence of drug resistance, it is currently the strongest known DNA methylation specific inhibitor, belonging to the S-phase cell cycle Specific drugs are suitable for the treatment of myelodysplastic syndrome (MDS). |
| preparation method | in argon environment, dissolve 2 '-deoxy- D-ribose (6.00g) in dry methanol (100 ml), slowly add hydrochloric acid (1.50 ml 1.25M hydrochloric acid in MeOH) to the reaction mixture, and stir the obtained solution at room temperature for 1 hour, pyridine (40 ml) was slowly added to the reaction system to neutralize the resulting solution. The solution was evaporated under vacuum to a nearly dry state, and the solution was co-evaporated with pyridine (15 ml). Dissolve the residue in pyridine (100 ml), then cool to 0 degrees, treat the residue with p-chlorobenzoyl chloride (107 mmol), stir the residue at room temperature for 18 hours, pour the slurry into a mixed system of water and dichloromethane (2:3,250 ml), use saturated sodium bicarbonate aqueous solution (2 x 20 ml), dilute sulfuric acid (2 x 20 ml) and water (2 x 20 ml) to wash the organic phase in turn to separate the organic layer, the resulting organic layer is dried with anhydrous magnesium sulfate, and the magnesium sulfate solid is removed by diatomite filtration, and the obtained filtrate is rotated and evaporated under vacuum to remove the reaction solvent, then dissolve the obtained slurry in a mixture of ether (45 ml) and acetic acid (60 ml), treat the solution with gaseous hydrochloric acid at 0 degrees for about 1 hour, and then wash the solid with cold ether The precipitate, and finally dry the precipitate under vacuum for 18 hours to obtain the target molecule 1-chloro-3, 5-di-p-chlorobenzoyloxy-2-deoxy-D-ribose. Fig. 1-synthesis route of chloro -3, 5-di-p-chlorobenzoyloxy-2-deoxy-D-ribose |
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: 1-Chloro-3,5-di(4-chlorbenzoyl)-2-deoxy-D-ribose Visit Supplier Webpage Request for quotationCAS: 3601-90-9
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 1-Chloro-3,5-di(4-chlorbenzoyl)-2-deoxy-D-ribose Request for quotation
CAS: 3601-90-9
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 3601-90-9
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: 1-Chloro-3,5-di(4-chlorbenzoyl)-2-deoxy-D-ribose Request for quotation
CAS: 3601-90-9
Tel: +86-21-56795779
Email: charles7788@worldyachem.com
Mobile: +86-13651600618
QQ: 2850607228
Wechat: 13651600618
WhatsApp: +8613651600618
CAS: 3601-90-9
Tel: +86-21-56795779
Email: charles7788@worldyachem.com
Mobile: +86-13651600618
QQ: 2850607228
Wechat: 13651600618
WhatsApp: +8613651600618
Product Name: 1-Chloro-3,5-di(4-chlorbenzoyl)-2-deoxy-D-ribose Visit Supplier Webpage Request for quotation
CAS: 3601-90-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 3601-90-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 1-Chloro-3,5-di(4-chlorbenzoyl)-2-deoxy-D-ribose Visit Supplier Webpage Request for quotationCAS: 3601-90-9
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 1-Chloro-3,5-di(4-chlorbenzoyl)-2-deoxy-D-ribose Request for quotation
CAS: 3601-90-9
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 3601-90-9
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: 1-Chloro-3,5-di(4-chlorbenzoyl)-2-deoxy-D-ribose Request for quotation
CAS: 3601-90-9
Tel: +86-21-56795779
Email: charles7788@worldyachem.com
Mobile: +86-13651600618
QQ: 2850607228
Wechat: 13651600618
WhatsApp: +8613651600618
CAS: 3601-90-9
Tel: +86-21-56795779
Email: charles7788@worldyachem.com
Mobile: +86-13651600618
QQ: 2850607228
Wechat: 13651600618
WhatsApp: +8613651600618
Product Name: 1-Chloro-3,5-di(4-chlorbenzoyl)-2-deoxy-D-ribose Visit Supplier Webpage Request for quotation
CAS: 3601-90-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 3601-90-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



