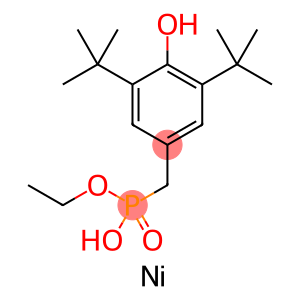
3,5-DI-TERT-BUTYL-4-HYDROXYBENZYLPHOSPHONICACID,MONOETHY.
Nickel bis[monoethyl(3,5-di-tert-butyl-4-hydroxylbenzyl)phosphonate]
CAS: 30947-30-9
Molecular Formula: C17H29NiO4P
3,5-DI-TERT-BUTYL-4-HYDROXYBENZYLPHOSPHONICACID,MONOETHY. - Names and Identifiers
3,5-DI-TERT-BUTYL-4-HYDROXYBENZYLPHOSPHONICACID,MONOETHY. - Physico-chemical Properties
| Molecular Formula | C17H29NiO4P |
| Molar Mass | 387.08 |
| Physical and Chemical Properties | It appears as a light yellow or light green powder depending on its moisture content. The melting point range is 180~200 ℃, the density is 1.10g/cm3, and the boiling point is 408.3 ℃ (1 atm). It is easily soluble in general organic solvents and has a solubility of about 5g/100mLH2O in water at 20 ℃. |
| Use | This product is a light stabilizer and antioxidant for polymer materials, with high stability to light and heat, good compatibility, extraction resistance, and low coloration. It has excellent stabilizing effects on fibers and films, mainly used for polyolefins, especially polypropylene fibers, films, and narrow bands (woven bags). It has a dyeing aid effect on polypropylene fibers. |
3,5-DI-TERT-BUTYL-4-HYDROXYBENZYLPHOSPHONICACID,MONOETHY. - Risk and Safety
| Toxicity | LD50 orl-rat: 3750 mg/kg NTIS** OTS0539888 |
3,5-DI-TERT-BUTYL-4-HYDROXYBENZYLPHOSPHONICACID,MONOETHY. - safety
Open Data Verified Data
This product has carcinogenicity, with an oral toxicity LD50 of 965mg/kg in mice. May cause skin and respiratory irritation to allergic individuals. In addition, toxic gases are generated during the decomposition of this product when heated. Therefore, it is necessary to pay attention to the ventilation of the operating environment, avoid inhaling the dust of this product, and store it against moisture and heat.
3,5-DI-TERT-BUTYL-4-HYDROXYBENZYLPHOSPHONICACID,MONOETHY. - Preparation method
Open Data Verified Data
1. React 3,5-di-tert-butylphenol with formaldehyde, dimethylamine, and solvent ethanol by heating to produce 3,5-di-tert-butylphenol-4-hydroxybenzyldimethylamine.
2. The above product reacts with diethyl phosphate in the presence of metallic sodium to produce 3,5-di-tert-butyl-4-hydroxybenzyl phosphate diethyl ester.
3. 35-, di-tert-butyl-4-hydroxybenzyl phosphate diethyl ester undergoes saponification reaction with sodium hydroxide to produce 3,5-di-tert-butyl-4-hydroxybenzyl phosphate monoethyl sodium salt.
4. The above products react with nickel chloride aqueous solution to produce this product.
3,5-DI-TERT-BUTYL-4-HYDROXYBENZYLPHOSPHONICACID,MONOETHY. - References
| EPA chemical substance information | information provided by: ofmpeb.epa.gov (external link) |
| production method | 1.3, 5-di-tert-butylphenol is added and reacted with formaldehyde, dimethylamine and ethanol as solvent, and heated to 75~80 ℃ with stirring to produce 3, 5-di-tert-butylphenol-4-hydroxybenzyldimethylamine 2. 3, 5-di-tert-butylphenol-4-hydroxybenzyldimethylamine is reacted with diethyl phosphate in the presence of metallic sodium at 110~130 ℃ to Form 3, diethyl 5-di-tert-butyl-4-hydroxybenzyl phosphate: 3. 3, 5-di-tert-butyl-4-hydroxybenzyl diethyl phosphate is saponified with sodium hydroxide at 125~135 ℃, formation-4-di-tert-butyl-4-hydroxybenzyl phosphoric acid monoethyl ester sodium salt: 4. The sodium salt of ethyl 3, 5-di-tert-butyl-4-hydroxybenzyl phosphate was reacted with an aqueous solution of nickel chloride at 40 °c, light stabilizer 2002 |
Supplier List
CAS: 30947-30-9
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 30947-30-9
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
2H-1-Benzopyran-3,5,7-triol,3,4-dihydro-2-(4-hydroxyphenyl)-, (2R,3R)-
4-dodecoxyaniline
(S)-Bunitrolol
2-(3-Amino-2,4,6-Triiodobenzyl)Butyric Acid
221466-41-7
AMINOPEPTIDASE M TYPE VI-S
二聚物
3,9-Dihydroxy-1,7-dimethyl-2,6-dibenzofurandicarboxylic acid
1353625-73-6
