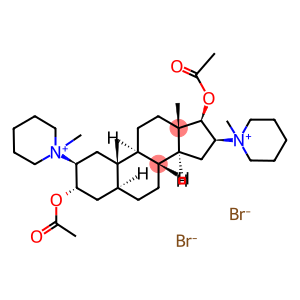
2beta,16beta-dipiperidino-5alpha-androstane-3alpha,17beta-dioldiacetatedimetho
pancuronium bromide
CAS: 15500-66-0
Molecular Formula: C35H60Br2N2O4
2beta,16beta-dipiperidino-5alpha-androstane-3alpha,17beta-dioldiacetatedimetho - Names and Identifiers
2beta,16beta-dipiperidino-5alpha-androstane-3alpha,17beta-dioldiacetatedimetho - Physico-chemical Properties
| Molecular Formula | C35H60Br2N2O4 |
| Molar Mass | 732.67 |
| Density | ~1, mp: 0°C |
| Melting Point | 215° |
| Boling Point | ~100°C |
| Solubility | Very soluble or freely soluble in water, very soluble in methylene chloride, freely soluble in ethanol (96 per cent). |
| Appearance | Off-white solid |
| Color | Clear, colorless solution |
| Odor | Odorless |
| Merck | 13,7077 |
| BRN | 4226892 |
| Storage Condition | 2-8°C |
| Physical and Chemical Properties | White or off-white crystalline or crystalline powder, slightly odorous, bitter taste. Hygroscopic. 1G dissolved in 30 parts of chloroform, 1 parts of water (20 degrees C). Soluble in water, ethanol, methanol, chloroform or dichloromethane, insoluble in ether. Acute toxicity LD50 mice (mg/kg):0.047 intravenous injection, 0.152 intraperitoneal injection, 0.167 subcutaneous injection, 21.9 oral. Acute toxicity LD50 Rat, rabbit (mg/kg):0.153,0.016 I. V. |
2beta,16beta-dipiperidino-5alpha-androstane-3alpha,17beta-dioldiacetatedimetho - Risk and Safety
| Hazard Symbols | Xn - Harmful |
| Risk Codes | 22 - Harmful if swallowed |
| UN IDs | UN 2811 6.1/PG 3 |
| WGK Germany | 3 |
| RTECS | TN4930000 |
| FLUKA BRAND F CODES | 10-21-33 |
| HS Code | 2933399090 |
| Hazard Class | 6.1(b) |
| Packing Group | III |
| Toxicity | LD50 in mice (mg/kg): 0.047 i.v.; 0.152 i.p.; 0.167 s.c.; 21.9 orally; in rats, rabbits: 0.153, 0.016 i.v. (Buckett, 1968) |
2beta,16beta-dipiperidino-5alpha-androstane-3alpha,17beta-dioldiacetatedimetho - Reference Information
| Muscle relaxants | Pancuronium bromide (pancuronium brom ide), the chemical name is dibromide (1, 1 ′-[3 α,17 β-Diacetoxy-5 α-androstane -2 β,16 β-diyl] bis [1-methylpiperidine]), it is a long-acting non-depolarizing muscle relaxant in the synthetic bisquaternary ammonium steroid. It is the earliest muscle relaxant used in the ICU. Its effect is 4-5 times stronger than that of rightline. All M choline receptors in the body have a blocking effect. The effect is similar to that of carrine chloride, but there are no side effects such as ganglion block and blood pressure drop, and it will not cause bronchospasm. It may also excite the heart β receptor or anti-vagal effect to increase the heart rate, increase blood pressure and increase Cardiac output. It is used as an auxiliary medicine for surgical or orthopedic anesthesia to obtain sufficient muscle relaxation. It can also be used for convulsive diseases such as tetanus. |
| Synthesis method | Using 5 α-androst-2-ene-17-one as raw material, the target compound Pancuronium bromide was synthesized through a 6-step reaction. Fig. 1 shows the synthesis route of pancuronium bromide |
| mechanism of action | after this product binds to N2 choline receptors on skeletal muscle cells, it does not produce depolarization, but competitively blocks the depolarization of the neurotransmitter acetylcholine, thus producing skeletal muscle relaxation effect. Its muscle relaxation efficiency is 3~5 times that of tuguoarine, and the effect is faster. This product cannot be taken orally, only for intravenous injection. 3~4 minutes after intravenous injection, the effect is obvious, and the maintenance time is about 20~30 minutes. About 30% of this product is metabolically inactivated in the liver, and most of it is excreted through the kidneys in its original form. Compared with tubuarine, this product is characterized by a moderate excitatory effect on the cardiovascular system, and no obvious histamine release effect. Continuous use has no accumulation and no adverse effects on the fetus. Neostigmine can resist the muscle relaxation effect of this product. |
| adverse reactions | can specifically block the cardiac atrioventricular node M choline receptor, with moderate vagus nerve effect, thus increasing heart rate, blood pressure, and even ventricular arrhythmia in individual patients. |
| taboo | hypertension, severe liver and kidney insufficiency and patients with obstructive jaundice should be used with caution. |
| precautions | 1. for patients with renal insufficiency and biliary obstruction, the duration is prolonged. 2. It has a moderate inhibitory effect on pseudo-cholinesterase activity, but it is relatively short-lived, and cholinesterase inhibitors have an antagonistic effect on it. 3. Repeated use has a savings effect. (2015-12-2) |
| Production method | The following compounds react with isopropenol acetate to carry out enol esterification, and then epoxidize p-chloroperbenzoic acid, hydrolyze potassium hydroxide, and make one of the epoxides ring to form a carbonyl group, then react with piperidine, sodium borohydride is reduced, acetylated, and finally reacted with two molecules of methyl bromide, and quaternized into salt to obtain pancuronium bromide. |
| toxic substance data | information provided by: pubchem.ncbi.nlm.nih.gov (external link) |
Last Update:2024-04-09 02:00:03
Supplier List
Spot supply
Product Name: Vecuronium Bromide Impurity 2(Vecuronium Bromide EP Impurity B) Visit Supplier Webpage Request for quotationCAS: 15500-66-0
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Spot supply
Product Name: Pancuronium dibromide Visit Supplier Webpage Request for quotationCAS: 15500-66-0
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Pancuronium bromide Request for quotation
CAS: 15500-66-0
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 15500-66-0
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Spot supply
Product Name: Vecuronium Bromide Impurity 2(Vecuronium Bromide EP Impurity B) Visit Supplier Webpage Request for quotationCAS: 15500-66-0
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Spot supply
Product Name: Pancuronium dibromide Visit Supplier Webpage Request for quotationCAS: 15500-66-0
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Pancuronium bromide Request for quotation
CAS: 15500-66-0
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 15500-66-0
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
View History
2beta,16beta-dipiperidino-5alpha-androstane-3alpha,17beta-dioldiacetatedimetho
PIPERIDINO(4-PIPERIDINYL)METHANONE
PIPERIDINO(4-PIPERIDINYL)METHANONE


