27353-36-2
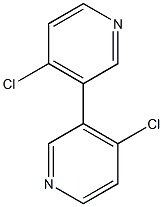
3,3'-Bipyridine, 4,4'-dichloro-
CAS: 27353-36-2
Molecular Formula: C10H6Cl2N2
27353-36-2 - Names and Identifiers
| Name | 3,3'-Bipyridine, 4,4'-dichloro- |
| Synonyms | 4,4'-dichloro-3,3'-dipyridine 4,4'-Dichloro-3,3'-bipyridine 4,4'-dichloro-3,3'-bipyridinyl 3,3'-Bipyridine, 4,4'-dichloro- 4-chloro-3-(4-chloropyridin-3-yl)pyridine |
| CAS | 27353-36-2 |
27353-36-2 - Physico-chemical Properties
| Molecular Formula | C10H6Cl2N2 |
| Molar Mass | 225.07 |
| Density | 1.363±0.06 g/cm3(Predicted) |
| Melting Point | 110℃ (hexane ) |
| Boling Point | 313.3±37.0 °C(Predicted) |
| pKa | 2.08±0.10(Predicted) |
| Storage Condition | under inert gas (nitrogen or Argon) at 2-8°C |
27353-36-2 - Introduction
3,3 '-Bipyridine, 4,4'-dichloro-is an organic compound with the chemical formula C10H6Cl2N2, which is a white crystalline solid. The following is a description of the nature, use, preparation and safety information of 3,3 '-Bipyridine, 4,4'-dichloro:
Nature:
-Appearance: White crystalline solid
-Solubility: Soluble in organic solvents such as ethanol, methanol and ether, insoluble in water
-Melting Point: about 130-133 ℃
-Stability: stable at room temperature, not easy to decompose
Use:
-catalyst: 3,3 '-Bipyridine, 4,4'-dichloro-is usually used as ligand of complex catalyst and participates in various organic reactions, such as coupling reaction, hydrogenation of olefins and aromatics, etc.
-Chemical Analysis: Can be used for the separation and determination of metal ions, such as platinum, iron, gold plasma detection.
Method:
3,3 '-Bipyridine, 4,4'-dichloro-can be prepared by the following methods:
-Synthesis of 4,4 '-Bipyridine from Pyridine
-Reaction of 4,4 '-Bipyridine with phosphorus chloride to generate 3,3'-Bipyridine, 4,4 '-dichloro-
Safety Information:
3,3 '-Bipyridine, 4,4'-dichloro-is an irritant substance that is irritating to the skin and eyes. Wear appropriate protective equipment during operation to avoid direct contact. At the same time, it should be operated in a well-ventilated place to avoid inhaling its dust. In case of accidental contact, rinse immediately with water and seek medical help. During storage, contact with oxidants, strong acids and strong bases should be avoided to avoid dangerous reactions.
Nature:
-Appearance: White crystalline solid
-Solubility: Soluble in organic solvents such as ethanol, methanol and ether, insoluble in water
-Melting Point: about 130-133 ℃
-Stability: stable at room temperature, not easy to decompose
Use:
-catalyst: 3,3 '-Bipyridine, 4,4'-dichloro-is usually used as ligand of complex catalyst and participates in various organic reactions, such as coupling reaction, hydrogenation of olefins and aromatics, etc.
-Chemical Analysis: Can be used for the separation and determination of metal ions, such as platinum, iron, gold plasma detection.
Method:
3,3 '-Bipyridine, 4,4'-dichloro-can be prepared by the following methods:
-Synthesis of 4,4 '-Bipyridine from Pyridine
-Reaction of 4,4 '-Bipyridine with phosphorus chloride to generate 3,3'-Bipyridine, 4,4 '-dichloro-
Safety Information:
3,3 '-Bipyridine, 4,4'-dichloro-is an irritant substance that is irritating to the skin and eyes. Wear appropriate protective equipment during operation to avoid direct contact. At the same time, it should be operated in a well-ventilated place to avoid inhaling its dust. In case of accidental contact, rinse immediately with water and seek medical help. During storage, contact with oxidants, strong acids and strong bases should be avoided to avoid dangerous reactions.
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: 4,4'-Dichloro-3,3'-bipyridine Visit Supplier Webpage Request for quotationCAS: 27353-36-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 3,3'-Bipyridine, 4,4'-dichloro- Request for quotation
CAS: 27353-36-2
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 27353-36-2
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: 4,4'-dichloro-3,3'-dipyridine Request for quotation
CAS: 27353-36-2
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 27353-36-2
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Product Name: 4,4'-dichloro-3,3'-dipyridine Visit Supplier Webpage Request for quotation
CAS: 27353-36-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 27353-36-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 4,4'-Dichloro-3,3'-bipyridine Visit Supplier Webpage Request for quotationCAS: 27353-36-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 3,3'-Bipyridine, 4,4'-dichloro- Request for quotation
CAS: 27353-36-2
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 27353-36-2
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: 4,4'-dichloro-3,3'-dipyridine Request for quotation
CAS: 27353-36-2
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 27353-36-2
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Product Name: 4,4'-dichloro-3,3'-dipyridine Visit Supplier Webpage Request for quotation
CAS: 27353-36-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 27353-36-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



