252670-82-9
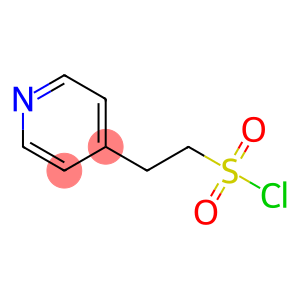
4-pyridineethanesulfonyl chloride
CAS: 252670-82-9
Molecular Formula: C7H8ClNO2S
252670-82-9 - Names and Identifiers
252670-82-9 - Physico-chemical Properties
| Molecular Formula | C7H8ClNO2S |
| Molar Mass | 205.66 |
| Density | 1.390 |
252670-82-9 - Introduction
chloride, chemical formula C8H10ClNO2S, is an organic compound. It is a colorless to slightly yellow liquid with a pungent odor at room temperature. The following is a description of its nature, use, preparation and safety information:
Nature:
-Appearance: Colorless to yellowish liquid
-Molecular weight: 209.69g/mol
-Solubility: Soluble in organic solvents such as chloroform, dichloromethane and acetone, almost insoluble in water
-Melting point:-2 ℃
-Boiling point: 82-84 ℃
-Density: 1.27 g/mL
Use: chloride is an important organic synthesis reagent, which is often used to synthesize functional groups in drugs and biomolecules.
-It can be used as a chlorination reagent to introduce sulfonyl chloride groups.
-It can also be used as an acylating reagent to introduce sulfonic acid groups into target molecules.
-In organic synthesis, it can be used to synthesize acid chloride derivatives, esters, acylating reagents, etc.
Method:
-the preparation method of chloride can be to react pyridine ethane sulfonamide with thionyl chloride (SO2Cl2) in the presence of ferrous chloride to obtain the desired product.
Safety Information: chloride is irritating and corrosive and should be avoided in contact with skin, eyes and respiratory tract.
-Wear appropriate personal protective equipment such as gloves, goggles and masks when in use.
-Pay attention to ventilation during operation and avoid inhalation of odors.
-In case of contact with skin or eyes, rinse immediately with plenty of water and seek medical help.
-should be sealed in storage, away from heat and open flame.
Nature:
-Appearance: Colorless to yellowish liquid
-Molecular weight: 209.69g/mol
-Solubility: Soluble in organic solvents such as chloroform, dichloromethane and acetone, almost insoluble in water
-Melting point:-2 ℃
-Boiling point: 82-84 ℃
-Density: 1.27 g/mL
Use: chloride is an important organic synthesis reagent, which is often used to synthesize functional groups in drugs and biomolecules.
-It can be used as a chlorination reagent to introduce sulfonyl chloride groups.
-It can also be used as an acylating reagent to introduce sulfonic acid groups into target molecules.
-In organic synthesis, it can be used to synthesize acid chloride derivatives, esters, acylating reagents, etc.
Method:
-the preparation method of chloride can be to react pyridine ethane sulfonamide with thionyl chloride (SO2Cl2) in the presence of ferrous chloride to obtain the desired product.
Safety Information: chloride is irritating and corrosive and should be avoided in contact with skin, eyes and respiratory tract.
-Wear appropriate personal protective equipment such as gloves, goggles and masks when in use.
-Pay attention to ventilation during operation and avoid inhalation of odors.
-In case of contact with skin or eyes, rinse immediately with plenty of water and seek medical help.
-should be sealed in storage, away from heat and open flame.
Last Update:2024-04-09 15:17:56
Supplier List
Product Name: 2-pyridin-4-ylethanesulfonyl chloride Request for quotation
CAS: 252670-82-9
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 252670-82-9
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Product Name: 2-pyridin-4-ylethanesulfonyl chloride Request for quotation
CAS: 252670-82-9
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 252670-82-9
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
View History
