2449-14-1
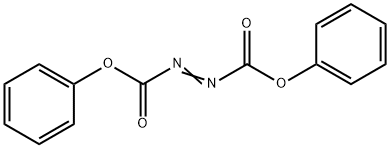
diphenyl azodicarboxylate
CAS: 2449-14-1
Molecular Formula: C14H10N2O4
2449-14-1 - Names and Identifiers
2449-14-1 - Physico-chemical Properties
| Molecular Formula | C14H10N2O4 |
| Molar Mass | 270.24 |
2449-14-1 - Introduction
Benzenediazodicarboxylate (diphenyl azodicarboxylate) is an organic compound. Its chemical formula is (C6H5N2CO2)2 and its molecular weight is 314.29.
diphenyl azodicarboxylate is a nitrogen-rich compound. Common physical properties include yellow solid or powdery crystals, insoluble in water, and soluble in many organic solvents such as ethanol and dimethylformamide.
diphenyl azodicarboxylate are mainly used as nitriding agents and activators. As a nitriding agent, it can initiate explosive reactions by reacting with some hydrogen-containing compounds and is often used in the preparation of explosives. As an activator, it can react with amine compounds to generate the corresponding diazonium salts. These diazonium salts have important applications in organic synthesis, such as the generation of amino radicals in nitrogen, and the amination and hydroxylation of organic chemical reactions.
The preparation method of diphenyl azodicarboxylate is generally obtained by electrochemical oxidation of aniline. First, aniline is dissolved in an appropriate amount of solvent as an electrolyte, a stabilizer is added to the electrolyte, and then the electrolytic cell is placed in the electrolytic cell, and the oxidation reaction is performed by electricity, and finally the diphenyl azodicarboxylate is obtained.
Regarding safety information, diphenyl azodicarboxylate is a highly unstable chemical that is explosive. It has an irritating effect on the skin, eyes and respiratory tract, and may cause poisoning by contact or inhalation. Therefore, when handling or handling the compound, appropriate personal protective measures must be taken, including wearing protective glasses, gloves and respirators. In addition, be careful to avoid contact with flammable materials or oxidants during storage and use to prevent fire or explosion. It is recommended to operate the compound under the guidance of an experienced person and to use appropriate safety facilities.
diphenyl azodicarboxylate is a nitrogen-rich compound. Common physical properties include yellow solid or powdery crystals, insoluble in water, and soluble in many organic solvents such as ethanol and dimethylformamide.
diphenyl azodicarboxylate are mainly used as nitriding agents and activators. As a nitriding agent, it can initiate explosive reactions by reacting with some hydrogen-containing compounds and is often used in the preparation of explosives. As an activator, it can react with amine compounds to generate the corresponding diazonium salts. These diazonium salts have important applications in organic synthesis, such as the generation of amino radicals in nitrogen, and the amination and hydroxylation of organic chemical reactions.
The preparation method of diphenyl azodicarboxylate is generally obtained by electrochemical oxidation of aniline. First, aniline is dissolved in an appropriate amount of solvent as an electrolyte, a stabilizer is added to the electrolyte, and then the electrolytic cell is placed in the electrolytic cell, and the oxidation reaction is performed by electricity, and finally the diphenyl azodicarboxylate is obtained.
Regarding safety information, diphenyl azodicarboxylate is a highly unstable chemical that is explosive. It has an irritating effect on the skin, eyes and respiratory tract, and may cause poisoning by contact or inhalation. Therefore, when handling or handling the compound, appropriate personal protective measures must be taken, including wearing protective glasses, gloves and respirators. In addition, be careful to avoid contact with flammable materials or oxidants during storage and use to prevent fire or explosion. It is recommended to operate the compound under the guidance of an experienced person and to use appropriate safety facilities.
Last Update:2024-04-10 22:29:15
Supplier List
Product Name: diphenyl azodicarboxylate Request for quotation
CAS: 2449-14-1
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 2449-14-1
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Product Name: diphenyl azodicarboxylate Request for quotation
CAS: 2449-14-1
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 2449-14-1
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
View History
