2229858-27-7
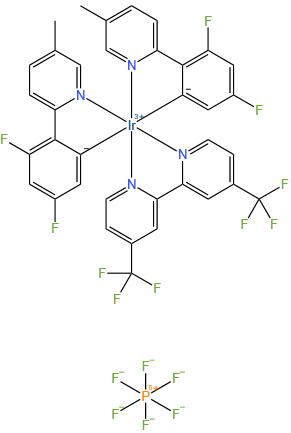
4,4'-Bis(trifluoromethyl)-2,2'-bipyridinebis[3,5-difluoro-2-[5-methyl-2-pyridinyl)phenyl] iridium(III) hexafluorophosphate
CAS: 2229858-27-7
Molecular Formula: C36H22F16IrN4P
2229858-27-7 - Names and Identifiers
| Name | 4,4'-Bis(trifluoromethyl)-2,2'-bipyridinebis[3,5-difluoro-2-[5-methyl-2-pyridinyl)phenyl] iridium(III) hexafluorophosphate |
| Synonyms | 4,4'-Bis(trifluoromethyl)-2,2'-bipyridinebis[3,5-difluoro-2-[5-methyl-2-pyridinyl)phenyl] iridium(III) hexafluorophosphate |
| CAS | 2229858-27-7 |
2229858-27-7 - Physico-chemical Properties
| Molecular Formula | C36H22F16IrN4P |
| Molar Mass | 1037.77 |
| Storage Condition | under inert gas (nitrogen or Argon) at 2–8 °C |
2229858-27-7 - Introduction
4,4 '-Bis(trifluoromethyl)-2,2'-bipyridinebis[3,5-difluoro-2-[5-methyll-2-pyridinyl) phenyl] iridium(III) hexafluorophosphate (abbreviated as BTMIrPF6) is an organic complex containing iridium. The following is a description of the nature, use, manufacture and safety information of the BTMIrPF6:
Nature:
-Appearance: The BTMIrPF6 is usually a yellow solid.
-Solubility: BTMIrPF6 can be dissolved in some organic solvents (such as chlorinated alkanes and ethers).
-Thermal stability: BTMIrPF6 has high thermal stability and can exist stably at high temperatures.
Use:
-Light-emitting material: BTMIrPF6 can be used as a light-emitting layer material of light-emitting devices such as organic light-emitting diodes (OLED), and has high light-emitting efficiency and light-emitting brightness.
-Photocatalyst: BTMIrPF6 can be used as a catalyst for photocatalytic reactions, such as photolysis of water to produce hydrogen.
-Organic synthesis: BTMIrPF6 catalysts that can be used for organic synthesis reactions, such as asymmetric hydrogenation of olefins.
Preparation Method:
The preparation method of the BTMIrPF6 includes the reaction of synthesizing BTMIr substrate and PF6-ion. Specific steps include:
1. Synthesis of BTMIR substrate: An appropriate amount of 4,4 '-bis (trifluoromethyl)-2,2'-bipyridine bis [3,5-difluoro -2-[5-methyl -2-pyridyl) phenyl] iridium complex is reacted with some excess deuterium trifluorocarboxylic acid to generate BTMIR substrate.
2. PF6-ion exchange: The BTMIr substrate is reacted with excess potassium hexafluorophosphate to form a BTMIrPF6 complex.
3. Crystallization Purification: The BTMIrPF6 product was purified by a crystallization purification step.
Safety Information:
- BTMIrPF6 have low toxicity under normal use conditions.
-Because iridium is a rare and expensive element, resource use and waste disposal should be properly managed when producing and disposing of BTMIrPF6 on an industrial scale.
-During operation, general laboratory safety procedures should be followed, including wearing appropriate personal protective equipment to avoid inhalation or contact with skin and eyes.
Nature:
-Appearance: The BTMIrPF6 is usually a yellow solid.
-Solubility: BTMIrPF6 can be dissolved in some organic solvents (such as chlorinated alkanes and ethers).
-Thermal stability: BTMIrPF6 has high thermal stability and can exist stably at high temperatures.
Use:
-Light-emitting material: BTMIrPF6 can be used as a light-emitting layer material of light-emitting devices such as organic light-emitting diodes (OLED), and has high light-emitting efficiency and light-emitting brightness.
-Photocatalyst: BTMIrPF6 can be used as a catalyst for photocatalytic reactions, such as photolysis of water to produce hydrogen.
-Organic synthesis: BTMIrPF6 catalysts that can be used for organic synthesis reactions, such as asymmetric hydrogenation of olefins.
Preparation Method:
The preparation method of the BTMIrPF6 includes the reaction of synthesizing BTMIr substrate and PF6-ion. Specific steps include:
1. Synthesis of BTMIR substrate: An appropriate amount of 4,4 '-bis (trifluoromethyl)-2,2'-bipyridine bis [3,5-difluoro -2-[5-methyl -2-pyridyl) phenyl] iridium complex is reacted with some excess deuterium trifluorocarboxylic acid to generate BTMIR substrate.
2. PF6-ion exchange: The BTMIr substrate is reacted with excess potassium hexafluorophosphate to form a BTMIrPF6 complex.
3. Crystallization Purification: The BTMIrPF6 product was purified by a crystallization purification step.
Safety Information:
- BTMIrPF6 have low toxicity under normal use conditions.
-Because iridium is a rare and expensive element, resource use and waste disposal should be properly managed when producing and disposing of BTMIrPF6 on an industrial scale.
-During operation, general laboratory safety procedures should be followed, including wearing appropriate personal protective equipment to avoid inhalation or contact with skin and eyes.
Last Update:2024-04-09 21:04:16
Supplier List
Spot supply
Product Name: 4,4'-Bis(trifluoromethyl)-2,2'-bipyridinebis[3,5-difluoro-2-[5-methyl-2-pyridinyl)phenyl] iridium(III) hexafluorophosphate Visit Supplier Webpage Request for quotationCAS: 2229858-27-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 4,4'-Bis(trifluoromethyl)-2,2'-bipyridinebis[3,5-difluoro-2-[5-methyl-2-pyridinyl)phenyl] iridium(III) hexafluorophosphate Request for quotation
CAS: 2229858-27-7
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 2229858-27-7
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Spot supply
Product Name: 4,4'-Bis(trifluoromethyl)-2,2'-bipyridinebis[3,5-difluoro-2-[5-methyl-2-pyridinyl)phenyl] iridium(III) hexafluorophosphate Visit Supplier Webpage Request for quotationCAS: 2229858-27-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 4,4'-Bis(trifluoromethyl)-2,2'-bipyridinebis[3,5-difluoro-2-[5-methyl-2-pyridinyl)phenyl] iridium(III) hexafluorophosphate Request for quotation
CAS: 2229858-27-7
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 2229858-27-7
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
View History

